Bausch (BHC) Starts Litigation Against Amneal for Xifaxan
Bausch Health BHC, along with its gastroenterology business, Salix Pharmaceuticals, Inc., announced that it has filed a lawsuit against Amneal Pharmaceuticals AMRX and its subsidiaries.
The lawsuit was filed on the heels of the Notice of Paragraph IV Certification, stating that Amneal submitted an abbreviated new drug application (ANDA) to the FDA seeking approval to market a generic version of Salix’s lead drug Xifaxan (rifaximin) 550 mg tablets.
Xifaxan 550 mg tablets are indicated for the reduction in risk of overt hepatic encephalopathy (HE) recurrence in adults and the treatment of irritable bowel syndrome with diarrhea (IBS-D) in adults.
Amneal asserts that certain patents listed in the FDA's Orange Book for Xifaxan are unenforceable, invalid and/or not infringed by Amneal's ANDA product.
The lawsuit initiates the litigation process under the Hatch-Waxman Act and triggers a 30-month stay of any potential FDA approval for Amneal's ANDA.
An unfavorable outcome will adversely impact sales. Xifaxan accounted for approximately 80% of the Salix reporting unit’s revenues in 2023, which came in at $2.2 billion.
The stock has risen 31% in the past six months compared with the industry’s growth of 26.2%.
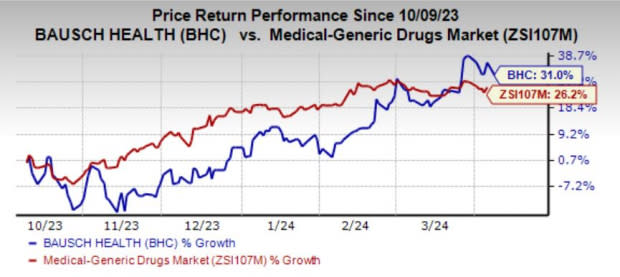
Image Source: Zacks Investment Research
We note that Bausch is already involved in litigation with Norwich Pharmaceuticals for Xifaxan.
In 2020, BHC and Alfasigma S.p.A received a Notice of Paragraph IV Certification from Norwich Pharmaceuticals Inc., whereby the latter asserted that the U.S. patents listed in the FDA’s Orange Book for Xifaxan are either invalid, unenforceable and/or will not be infringed by the commercial manufacture, use or sale of Norwich’s generic rifaximin tablets, 550 mg, for which an ANDA has already been filed by Norwich.
Thereafter, a lawsuit was filed against Norwich, alleging infringement of one or more claims of the Xifaxan patents, thereby triggering a 30-month stay of the approval of Norwich’s ANDA.
The court issued a final judgment in August 2022 after finding that the patents protecting the use of Xifaxan tablets for the reduction in risk of HE recurrence were valid and infringed and the patents protecting the composition and use of Xifaxan for treating IBS-D invalid. This judgment will prevent the FDA approval of Norwich’s ANDA until October 2029.
BHC appealed the decision and consequently Norwich claimed to have removed the HE indication from its existing ANDA and then filed a motion in the District Court requesting modification of the FDA’s decision.
BHC opposed the motion. The District Court denied Norwich’s motion and confirmed that the FDA remained enjoined from granting final approval to Norwich’s ANDA until October 2029. Norwich filed its appeal to the U.S. Court of Appeals for the Federal Circuit in May 2023. BHC awaits a decision from the Federal Circuit.
The FDA granted tentative approval to Norwich’s ANDA in June 2023, but confirmed that it is enjoined from granting final approval until October 2029. Norwich has filed a lawsuit against the FDA for the same and BHC has intervened it.
Bausch had previously received Paragraph IV Certifications for Xifaxan tablets from Teva Pharmaceuticals, Sandoz Inc. and Sun Pharmaceuticals. The company has settled the matters with these companies.
Zacks Rank and Stocks to Consider
Bausch currently carries a Zacks Rank #3 (Hold).
A couple of better-ranked stocks from the biotech sector are ADMA Biologics, Inc. ADMA and ANI Pharmaceuticals, Inc. ANIP, both sporting a Zacks Rank #1 (Strong Buy) at present. You can see the complete list of today’s Zacks #1 Rank stocks here.
In the past 60 days, estimates for ADMA Biologics’ 2024 earnings per share (EPS) have improved from 22 to 30 cents. In the past year, shares of ADMA have surged 90.9%.
ADMA Biologics’ earnings beat estimates in three of the trailing four quarters and met once, delivering an average surprise of 85.00%.
In the past 60 days, estimates for ANI Pharmaceuticals’ 2024 EPS have improved from $4.06 to $4.43. In the past year, shares of ANIP have surged 75.7%.
The company’s earnings beat estimates in each of the trailing four quarters, delivering an average surprise of 109.06%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
ANI Pharmaceuticals, Inc. (ANIP) : Free Stock Analysis Report
ADMA Biologics Inc (ADMA) : Free Stock Analysis Report
Bausch Health Cos Inc. (BHC) : Free Stock Analysis Report
AMNEAL PHARMACEUTICALS, INC. (AMRX) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 