Biotech Stock Roundup: BIIB's Q1 Results, ARWR Up on Data, INCY & BLUE Offer Updates
The biotech sector has been in focus in the past week with key pipeline and regulatory updates. Among these, Biogen BIIB reports first-quarter results while Incyte INCY gets FDA approval for novel cream formulation of JAK1/JAK2 inhibitor ruxolitinib.
Recap of the Week’s Most Important Stories:
Q1 Results From Biogen: Biogen reported first-quarter 2023 adjusted earnings per share (EPS) of $3.40, beating the Zacks Consensus Estimate of $3.25 but down from $3.62 reported a year ago. Earnings declined 6% year over year due to lower revenues. Total revenues came in at $2.46 billion, down 3% on a reported basis (flat on a constant-currency basis) from the year-ago quarter, hurt by lower sales of multiple sclerosis (MS) drugs like Tecfidera and Vumerity. Sales beat the Zacks Consensus Estimate of $2.34 billion. The company reaffirmed its previously-issued financial outlook for 2023. The company also announced that it has deprioritized certain programs in stroke, gene therapy and ophthalmology as part of ongoing R&D pipeline optimization.
Concurrently, Biogen announced that the FDA has approved tofersen100 mg/15mL injection under the brand name Qalsody for the treatment of amyotrophic lateral sclerosis (ALS) in adults who have a mutation in the superoxide dismutase 1 (SOD1) gene. This indication is approved under accelerated approval based on a reduction in plasma neurofilament light chain (NfL) observed in patients treated with Qalsody.
Biogen currently has a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Incyte’s Drug Approval: Incyte announced that the European Commission (“EC”) approved the novel cream formulation of JAK1/JAK2 inhibitor ruxolitinib (15 mg/g), Opzelura, for the treatment of non-segmental vitiligo with facial involvement in adults and adolescents from 12 years of age.The EC approval was based on data from two pivotal phase III studies (TRuE-V1 [NCT04052425] and TRuE-V2 [NCT04057573]), evaluating the efficacy and the safety of Opzelura versus vehicle (non-medicated cream) in more than 600 people with non-segmental vitiligo, age 12 and older. Results from the TRuE-V program showed that treatment with Opzelura led to significant improvements in facial and total body repigmentation versus vehicle.
EC’s approval was in the cards, as the European Medicines Agency’s Committee for Medicinal Products for Human Use had earlier given a positive opinion on the same. Opzelura is already approved in the United States for the topical treatment of non-segmental vitiligo in patients 12 years of age and older, and for the topical short-term and non-continuous chronic treatment of mild to moderate atopic dermatitis.
Arrowhead Surges on Data: Shares of Arrowhead Pharmaceuticals Inc. ARWR surged after it announced interim results from an ongoing phase I/II clinical study of ARO-RAGE, the company’s investigational RNA interference (RNAi) therapeutic designed to reduce the production of the receptor for advanced glycation end products (RAGE) as a potential treatment for inflammatory pulmonary diseases, such as asthma.
Interim results showed up to 90% serum sRAGE reduction with a mean maximum reduction of 80% after two doses. The mean maximum reductions at 10 mg to 44 mg dose levels showed a dose response ranging from 31% to 59% The duration of the pharmacologic effect persisted for at least six weeks after the second administration of the 92 mg dose with further follow-up ongoing. The reductions in sRAGE as measured in bronchoalveolar lavage fluid (BALF) at day 31 after a single dose showed a mean reduction at 92 mg dose was 75% with a maximum decrease of 92% and mean reductions at 10 to 44 mg doses ranging from 44% to 52% Arrowhead will provide additional information in June.
Morphic Reports Ulcerative Colitis Data: Morphic Therapeutic MORF announced positive top-line data from the main cohort of the open-label EMERALD-1 phase IIa study of MORF-057, an oral small molecule inhibitor of the α4β7 integrin, in adults with moderate to severe ulcerative colitis (UC). MORF-057 achieved the primary endpoint, demonstrating a statistically significant reduction in the robarts histopathology index (RHI) score of 6.4 points (p=0.002) from baseline to week 12. MORF-057 achieved 25.7% clinical remission by modified mayo clinic score (mMCS). The candidate was generally well tolerated at the dose of 100 mg BID (twice daily) with no serious adverse events and no safety signal. Shares of Morphic were up on the same.
Regulatory Update From bluebird: bluebird bio BLUE announced that it submitted its biologics license application (BLA) to the FDA for lovotibeglogene autotemcel (lovo-cel) gene therapy in patients with sickle cell disease (SCD) ages 12 and older who have a history of vaso-occlusive events (VOEs). The BLA includes a request for Priority Review, which, if granted, would shorten the FDA’s review of the application to six months from the time of filing versus a standard review timeline of 10 months. The BLA submission is based on efficacy results from 36 patients in the HGB-206 Group C cohort with a median of 32 months of follow-up and two patients in the HGB-210 study with 18 months of follow-up each. The BLA submission also includes safety data from 50 patients treated across the entire lovo-cel program, including six patients with six or more years of follow-up. A potential approval will make lovo-cel -bluebird bio’s third ex-vivo gene therapy approved by the FDA for a rare genetic disease.
Performance
The Nasdaq Biotechnology Index has lost 0.85% in the past five trading sessions. Among the biotech giants, Moderna has lost 5.42% during the period. Over the past six months, shares of GSK have soared 16.69%. (See the last biotech stock roundup here: Biotech Stock Roundup: MDGL, PCVX Up on Study Data, MRNA Provides Updates)
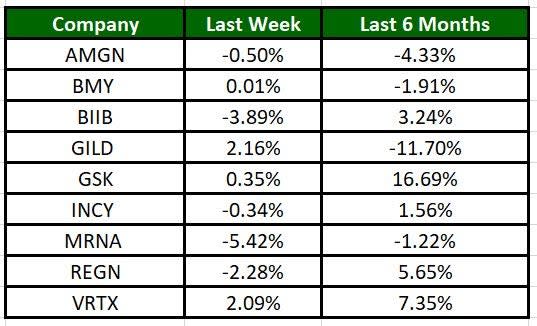
Image Source: Zacks Investment Research
What's Next in Biotech?
Stay tuned for more earnings and pipeline updates.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Biogen Inc. (BIIB) : Free Stock Analysis Report
Incyte Corporation (INCY) : Free Stock Analysis Report
Arrowhead Pharmaceuticals, Inc. (ARWR): Free Stock Analysis Report
bluebird bio, Inc. (BLUE) : Free Stock Analysis Report
Morphic Holding, Inc. (MORF) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 