Clovis (CLVS) Q2 Loss Wider Than Expected, Revenues Miss
Clovis Oncology CLVS reported a net loss of 50 cents per share in second-quarter 2022, narrower than the year-ago period’s loss of 61 cents.
Adjusted loss (excluding non-cash adjustment of $9.7 million in other manufacturing costs related to the expected expiration of Rubraca currently in inventory) was 43 cents per share, wider than the Zacks Consensus Estimate of a loss of 42 cents. In the year-ago quarter, the company posted an adjusted loss of 61 cents.
Net revenues — entirely from Clovis’ only marketed drug PARP inhibitor Rubraca — were down 13% year over year to $32.1 million, missing the Zacks Consensus Estimate of $37.8 million.
Quarter in Detail
Sales of Rubraca in the United States were $22.7 million, down 18.1% year over year. Ex-U.S. market sales were $9.4 million for the second quarter, up 3.3% year over year. Lower sales in the U.S. were due to COVID-19 impacts as fewer patients were treated for ovarian cancer in the second-line setting amid the pandemic. Overall, Rubraca sales also declined 6% quarter over quarter.
For the second quarter, research & development expenses decreased 20% year over year to $36.4 million, primarily owing to lower spending on Rubraca clinical studies.
Selling, general and administrative expenses dropped 1% year over year to $32.6 million.
Clovis ended the quarter with $94.6 million of cash equivalents and available-for-sale securities compared with $122.2 million on Mar 31, 2022.
CLVS also faces the risk of a severe cash crunch. The company announced the need to raise additional capital to support its operations beyond February 2023. Shares of Clovis fell 12.1% in pre-market trading on Aug 8 following this announcement.
The stock has declined 38.8% in the year compared with the industry’s 18.0% decrease.
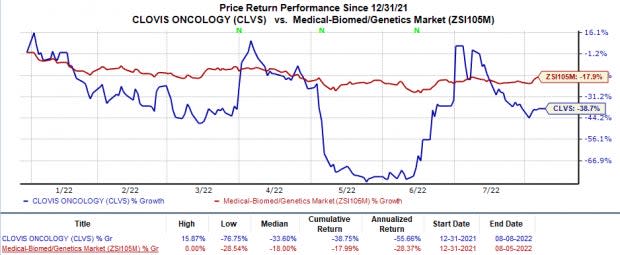
Image Source: Zacks Investment Research
Pipeline Updates
Label Expansion Studies on Rubraca
Rubraca is currently approved for treating certain patients with ovarian and prostate cancer. Several label expansion studies are ongoing on Rubraca.
The phase III ATHENA study is evaluating Rubraca as a monotherapy and in combination with Bristol Myers’ BMY Opdivo for advanced ovarian cancer as a first-line maintenance treatment.
In March 2022, Clovis reported positive top-line data from the monotherapy arm of the ATHENA study (ATHENA-MONO), which achieved its primary endpoint of a statistically significant progression-free survival (PFS) over placebo. Based on this data, CLVS intends to seek label expansion for Rubraca as a first-line ovarian cancer maintenance treatment in both the United States and Europe in third-quarter 2022.
Top-line data from the combination arm of the ATHENA study (ATHENA-COMBO) is anticipated in first-quarter 2023. The ATHENA study is part of a broader clinical collaboration with Bristol Myers, finalized in 2017. Opdivo is one of the key drivers of Bristol Myers’ top line. During second-quarter 2022, Bristol Myers recorded $2.1 billion from Opdivo sales.
A confirmatory phase III TRITON3 study is evaluating Rubraca in metastatic castration-resistant prostate cancer (mCRPC) patients with tumors associated with BRCA mutations and ATM mutations. Data from this study is now expected in fourth-quarter 2022, a delay by one quarter. This will serve as a confirmatory study for the continued approval of Rubraca to treat mCRPC. It will also serve as a potential second-line label expansion for Rubraca to address mCRPC.
FAP-2286
Clovis is currently evaluating FAP-2286, its lead peptide-targeted radionuclide therapy (PTRT) and imaging agent candidate, in phase I/II LuMIERE study across multiple tumor types.
In June 2022, management reported initial data from the phase I portion of the LuMIERE study, which showed preliminary evidence of activity of FAP-2286 as a treatment and imaging agent across a wide range of solid tumors. The company intends to use this data to start the phase II portion of the study in fourth-quarter 2022. Presently, CLVS is also enrolling participants in the third dose cohort of this study.
Management is currently engaged in discussions with potential partners to license the development and commercial rights for FAP-2286. This will help the company to meet its liquidity needs and continue as a going concern.
Clovis Oncology, Inc. Price
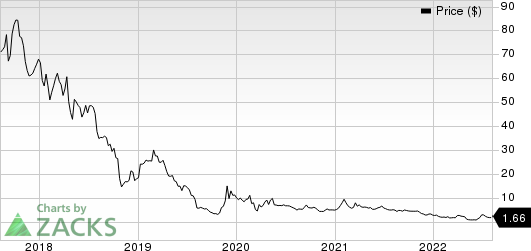
Clovis Oncology, Inc. price | Clovis Oncology, Inc. Quote
Zacks Rank & Other Stocks to Consider
Clovis currently carries a Zacks Rank #3 (Hold). Other top-ranked stocks in the overall healthcare sector include Alkermes ALKS and Novavax NVAX, each carrying a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Alkermes’ stock has risen 10.8% this year so far. Alkermes’ estimates for 2022 have gone up from a loss of 17 cents per share to earnings of 20 cents per share, while the consensus estimate for 2023 earnings has increased from 31 cents per share to 50 cents per share in the past 30 days.
Alkermes beat earnings estimates in each of the last four quarters, delivering an average earnings surprise of 325.48%, on average. In the last reported quarter, ALKS reported an earnings surprise of 50.00%.
Novavax’s stock has plunged 57.9% this year so far. Novavax’s earnings estimates for 2023 have increased from $10.43 per share to $10.62 per share over the past 30 days.
Novavax missed earnings estimates in each of the last four quarters, delivering a negative earnings surprise of 184.49%, on average. In the last reported quarter, NVAX reported a negative earnings surprise of 23.12%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Bristol Myers Squibb Company (BMY) : Free Stock Analysis Report
Alkermes plc (ALKS) : Free Stock Analysis Report
Novavax, Inc. (NVAX) : Free Stock Analysis Report
Clovis Oncology, Inc. (CLVS) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 