Erasca (ERAS) Inks Deal With Pfizer for Colorectal Cancer Study
Erasca, Inc. ERAS announced that it has entered into a collaboration and supply agreement with pharma giant Pfizer Inc. PFE to support a clinical study. Per reports, the study will evaluate the company’s oral ERK1/2 inhibitor ERAS-007 in combination with the latter’s BRAF inhibitor Braftovi (encorafenib) and Eli Lilly’s LLY EGFR inhibitor Erbitux (cetuximab) to treat patients with BRAF V600E-mutant metastatic colorectal cancer (mCRC). The study is expected to begin in the second half of 2021.
Shares of Erasca were down 10.8% on Wednesday following the aforementioned announcement. The stock has inched up 0.1% in the past month compared with the industry’s rise of 0.5%.
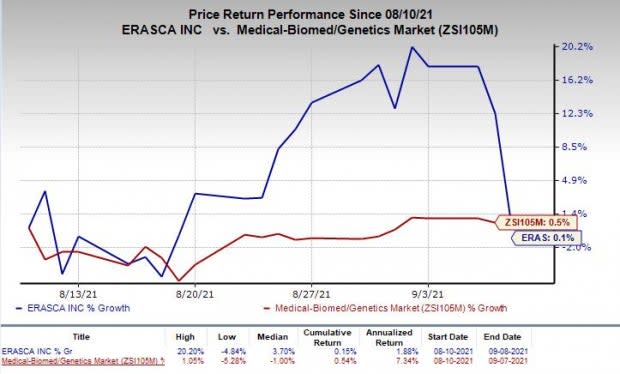
Image Source: Zacks Investment Research
The deal will support a clinical proof-of-concept study wherein Erasca will provide funding for the study and Pfizer will supply Braftovi while both companies will jointly review the study results.
We note that the combo of Braftovi plus Erbitux is currently approved to address previously-treated patients with BRAF V600E-mutant mCRC.
With the above-mentioned agreement, Erasca is looking to explore whether the ERK inhibition with ERAS-007 in combination with Braftovi plus Erbitux is able to reduce the emergence of resistance and improve the treatment benefits in BRAF V600E-mutant mCRC.
Apart from the HERKULES-3 study, ERAS-007 is being evaluated in various early-to mid-stage studies, both as a monotherapy and a combo regimen for treating multiple tumor types.
The phase Ib/II HERKULES-1 study is evaluating ERAS-007 as a single agent and in combination with Erasca’sSHP2 inhibitor ERAS-601 for treating advanced solid tumors. The phase Ib/II HERKULES-2 study is investigating ERAS-007 in combination with various agents in patients with non-small cell lung cancer. The phase Ib/II HERKULES-4 study is evaluating ERAS-007 in combination with various agents for addressing hematologic malignancies.
We remind investors that Erasca started trading on the NASDAQ stock exchange following its initial public offering in July, 2021.
Zacks Rank & Stock to Consider
Erasca currently carries a Zacks Rank #3 (Hold). A better-ranked stock in the biotech sector is Spero Therapeutics, Inc. SPRO, which has a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Spero Therapeutics’ loss per share estimates have narrowed 8.2% for 2021 and 10.6% for 2022 over the past 60 days.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Pfizer Inc. (PFE) : Free Stock Analysis Report
Eli Lilly and Company (LLY) : Free Stock Analysis Report
Spero Therapeutics, Inc. (SPRO) : Free Stock Analysis Report
Erasca, Inc. (ERAS) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 