Global $250+ Bn Pharmaceutical API Manufacturing (Chemical & Biological) Markets, 2016-2021, 2021-2026F, 2031F

Global Pharmaceutical API Manufacturing Market
Dublin, April 13, 2022 (GLOBE NEWSWIRE) -- The "Pharmaceutical API Manufacturing Global Market Report 2022: By Therapy, By API, By Drug" report has been added to ResearchAndMarkets.com's offering.
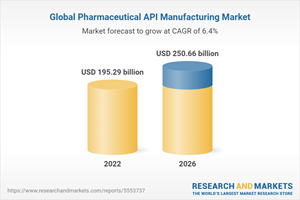
The global pharmaceutical API manufacturing market is expected to grow from $179.05 billion in 2021 to $195.29 billion in 2022 at a compound annual growth rate (CAGR) of 9.1%.
The growth is mainly due to the companies resuming their operations and adapting to the new normal while recovering from the COVID-19 impact, which had earlier led to restrictive containment measures involving social distancing, remote working, and the closure of commercial activities that resulted in operational challenges. The market is expected to reach $250.66 billion in 2026 at a CAGR of 6.4%.
The pharmaceutical active pharmaceutical ingredients (API) manufacturing market consists of sales of pharmaceutical active pharmaceutical ingredients and related services. Active pharmaceutical ingredients are used to produce drugs which treat various diseases such as cardiovascular diseases, rare genetic disorders, infections, metabolic disorders, cancers, respiratory diseases and others. Active pharmaceutical ingredient is a chemical which is responsible for the pharmacological activity in the living body and are used as a major base for preparation of drugs.
The main therapy areas of pharmaceutical API manufacturing are cardiovascular disorders, metabolic disorders, neurological disorders, oncology, musculoskeletal disorders, NSAIDs and other therapeutics uses. Cardiovascular diseases are a group of heart and blood vessel disorders. The various API type include chemical API and biological API that are used to produce innovative drugs, generic prescription, over-the-counter (OTC) drugs.
North America was the largest region in the pharmaceutical API manufacturing market in 2021. Middle East is expected to be the fargest growing region in the forecast period. The regions covered in this report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East and Africa.
The rise in aging population is one of the major drivers of the pharmaceutical API manufacturing market. As the aging population increases, the demand for pharmaceutical drugs also increases. API acts as a specialty drug in these medicines to cure a particular disease. For instance, in 2019, number of people over the age of 65 were 703 million, in 2050, the number of elderly people is expected to double to 1.5 billion. This led to increase demand for pharmaceutical drugs, significantly impacting the pharmaceutical API manufacturing market growth.
Stringent regulations for approval of pharmaceutical drugs acts as a restraint on the pharmaceutical API manufacturing market. Various regions have their own set of rules, guidelines, and regulations set by their respective drugs regulatory bodies. For example, in Japan, the manufacturers have to register an API with Japan's Pharmaceuticals and Medical Devices Agency (PMDA) through the country's Drug Master File (DMF) system.
Products like APIs and new excipients i.e. inactive substances used in a drug should be registered compulsorily. The API manufacturer is also responsible for DMF registration, even if they don't have their office in Japan. They should appoint an in-country caretaker (ICC) to apply on their behalf.
Also, if there are any changes to be made to DMF products, either an application for change in registration or a minor change notification should be submitted by the ICC to the PMDA. This whole process delays the drugs approvals, thereby negatively affecting the pharmaceuticals API manufacturing market.
Many companies use modern technologies such as process control, and powder handling to optimize API manufacturing. These technologies include advances in Lyophilization i.e. freeze-drying process. It is a low-temperature dehydration process which involves freezing the product, lowering pressure and then removing the ice formed.
Key Topics Covered:
1. Executive Summary
2. Pharmaceutical API Manufacturing Market Characteristics
3. Pharmaceutical API Manufacturing Market Trends And Strategies
4. Impact Of COVID-19 On Pharmaceutical API Manufacturing
5. Pharmaceutical API Manufacturing Market Size And Growth
5.1. Global Pharmaceutical API Manufacturing Historic Market, 2016-2021, $ Billion
5.1.1. Drivers Of The Market
5.1.2. Restraints On The Market
5.2. Global Pharmaceutical API Manufacturing Forecast Market, 2021-2026F, 2031F, $ Billion
5.2.1. Drivers Of The Market
5.2.2. Restraints On the Market
6. Pharmaceutical API Manufacturing Market Segmentation
6.1. Global Pharmaceutical API Manufacturing Market, Segmentation By Therapy Area, Historic and Forecast, 2016-2021, 2021-2026F, 2031F, $ Billion
Cardiovascular disorders
Metabolic disorders
Neurological disorders
Oncology
Musculoskeletal disorders
NSAIDs
Other therapeutics uses
6.2. Global Pharmaceutical API Manufacturing Market, Segmentation By API Type, Historic and Forecast, 2016-2021, 2021-2026F, 2031F, $ Billion
Chemical API
Biological API
6.3. Global Pharmaceutical API Manufacturing Market, Segmentation By Drug Type, Historic and Forecast, 2016-2021, 2021-2026F, 2031F, $ Billion
Innovative Drugs
Generic Prescription
Over-the-counter (OTC) Drugs
7. Pharmaceutical API Manufacturing Market Regional And Country Analysis
7.1. Global Pharmaceutical API Manufacturing Market, Split By Region, Historic and Forecast, 2016-2021, 2021-2026F, 2031F, $ Billion
7.2. Global Pharmaceutical API Manufacturing Market, Split By Country, Historic and Forecast, 2016-2021, 2021-2026F, 2031F, $ Billion
Companies Mentioned
Teva Pharmaceutical Industries Ltd.
Sun Pharmaceutical Industries Ltd.
Boehringer Ingelheim
Pfizer inc.
Aurobindo Pharma
Novartis AG
Dr. Reddy's Laboratories
Merck & Co. inc.
BASF SE.
Bayer Healthcare
Mylan N.V.
Albemarle Corporation
Actavis plc.
Hospira inc.
Ranbaxy Laboratories
Virbac
Zoetis
Heska Co.
AbbVie inc.
AstraZeneca plc.
Eli Lilly and Company
GlaxoSmithKline plc.
Sanofi
Bristol-Myers Squibb
Cipla inc.
F.Hoffmann-La Roche Ltd.
For more information about this report visit https://www.researchandmarkets.com/r/ddd51c
Attachment
CONTACT: CONTACT: ResearchAndMarkets.com Laura Wood, Senior Press Manager press@researchandmarkets.com For E.S.T Office Hours Call 1-917-300-0470 For U.S./CAN Toll Free Call 1-800-526-8630 For GMT Office Hours Call +353-1-416-8900


 Yahoo Finance
Yahoo Finance 
