Global $72 Billion Point of Care & Rapid Diagnostics Markets, 2022-2027 - Increasing Provider Awareness About Novel Technologies

Global Point of Care and Rapid Diagnostics Market
Dublin, April 22, 2022 (GLOBE NEWSWIRE) -- The "Global Point of Care & Rapid Diagnostics Market by Product (Glucose, Infectious Disease (HIV, Hepatitis C), Pregnancy Test), Platform (Microfluidics, Dipsticks), Mode of Purchase (OTC, Prescription), End User (Pharmacy, Hospital, Home) - Forecast to 2027" report has been added to ResearchAndMarkets.com's offering.
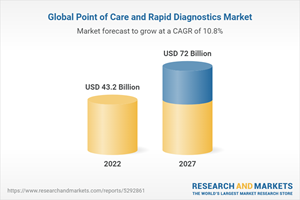
The global point of care diagnostics market is expected to reach USD 72.0 billion by 2027 from an estimated USD 43.2 billion in 2022, at a CAGR of 10.8% from 2022 to 2027.
Point-of-care testing (POCT) includes screening or diagnostic tests performed outside traditional laboratories. These tests are designed to be used at or near the site where the patient is located (such as at home, in an ambulance, at a physician's office, and other locations).
POCT allows rapid and reliable diagnostic testing through which outcomes can be obtained instantly, helping physicians/patients to make decisions related to care remotely and as soon as possible. Market growth is mainly driven by the high prevalence of infectious diseases, supportive government policies and rising number of CLIA-waived POC tests.
In addition, healthcare decentralization and greater investments and funding for product development are expected to offer growth opportunities for players in the point of care diagnostics market. POC tests can greatly improve the management of infectious diseases, especially in developing countries where access to timely medical care is a challenge and the healthcare infrastructure is antiquated and sparse.
The high prevalence of infectious diseases, coupled with the inadequate healthcare infrastructural facilities in developing countries, is expected to drive the adoption of POC testing.
The Glucose Monitoring Products segment accounted for the largest share of the point of care diagnostics market, by product type, in 2021
Based on products, the point-of-care diagnostics market is segmented into glucose monitoring products, cardiometabolic monitoring products, COVID-19 testing products, infectious disease testing products, coagulation monitoring products, pregnancy & fertility testing products, hematology testing products, tumor/cancer marker testing products, fecal occult testing products, urinalysis testing products, drug-of-abuse testing products, cholesterol testing products, and other products.
In 2021, the glucose monitoring products segment accounted for the largest share of the point-of-care diagnostics market. This can be attributed to the growing diabetic patient population base, ongoing technological advancements, and the growing preference for home glucose testing.
The Lateral Flow Assays segment accounted for the largest share of the point of care diagnostics market, by platform, in 2021
Based on platform, the point-of-care diagnostics market is segmented into lateral flow assays, immunoassays, microfluidics, dipsticks, and molecular diagnostics. The lateral flow assays segment accounted for the largest share of the market in 2021. The widespread adoption of lateral flow assays in POC testing as a replacement for lengthy, conventional laboratory procedures is a key factor contributing to the growth of this segment.
The OTC Testing Products segment, by mode of purchase, accounted for the largest share of the global point of care diagnostics market in 2021
On the basis of mode of purchase, the point-of-care diagnostics market is segmented into OTC testing products and prescription-based testing products.
The OTC testing products segment accounted for the largest share of the market in 2021. The growing prevalence of lifestyle diseases and the favorable reimbursement scenario for OTC testing products are the major factors driving the growth of this market segment.
On the basis of End Users, Hospitals & Critical Care Centers segment accounted for the largest share of the global point of care diagnostics market, in 2021
The hospitals & critical care centers segment is expected to dominate the market during the forecast period, owing to the increasing adoption of POC devices offering reduced turnaround time. The clinical laboratories segment accounted for the largest share of the point-of-care diagnostics market in 2021.
However, the home care and self-testing segment is projected to register the highest CAGR during the forecast period, owing to the increasing prevalence of chronic diseases (requiring long-term care and frequent monitoring), growing awareness about home care, and the increasing availability of user-friendly and advanced POC diagnostic products.
The Latin America market is expected to grow at the highest CAGR during the forecast period
In 2021, North America accounted for the largest share of this market, followed by Europe. The rising prevalence of lifestyle diseases, growing number of product approvals, and government initiatives to support the wider adoption of POC testing products are the key factors driving the growth of the North American point-of-care diagnostics market.
On the other hand, the Latin American market is projected to register the highest growth during the forecast period due to government efforts to increase awareness about the early detection of diseases and regular health check-ups, further promoting point-of-care devices.
As of 2020, the point of care diagnostics market is dominated by Roche Diagnostics (Switzerland), Abbott Laboratories (US), Siemens Healthineers (Germany), Danaher Corporation (US), Quidel Corporation (US). Other leading players are Becton, Dickinson, and Company (US), Biomeriux S.A. (France), and Chembio Diagnostics (US).
Market Dynamics
Drivers
High Prevalence of Infectious Diseases
Increasing Incidence of Target Conditions
Supportive Government Policies
Dearth of Skilled Laboratory Technicians
Rising Number of Clia-Waived Poc Tests
Restraints
Pricing Pressure Owing to Reimbursement Cuts and Budget Constraints
Stringent Regulatory Policies
Opportunities
Emerging Markets
Healthcare Decentralization
Increasing Provider Awareness About Novel Technologies
Poc Tests with Multiplexing Capabilities
Growing Investments and Funding for Product Development
Challenges
Lack of Alignment with Definitive Central Lab Methods
Inadequate Adoption of Poc Devices in Professional Settings
Reluctance to Change Existing Diagnostic Practices
Premium Pricing of Novel Platforms
Technology Analysis: Emerging Point-Of-Care Applications
Sepsis Biomarkers
Stroke/Cardiac Markers
Thyroid Testing
Dna Testing
Endocrine Testing
Respiratory Diagnostics
Regulatory Landscape
Industry Trends Related to COVID-19
Value Chain Analysis
Supply Chain Analysis
Prominent Companies
Small & Medium-Sized Enterprises
End-users
Pricing Trend Analysis
Porter's Five Forces
Company Profiles
Key Players
Abbott
Siemens Healthineers Ag
Quidel Corporation
Danaher Corporation
Becton, Dickinson and Company
Thermo Fisher Scientific
Biomerieux S.A.
Chembio Diagnostics, Inc.
Ekf Diagnostics
Trinity Biotech plc
Instrumentation Laboratory (A Werfen Company)
Nova Biomedical
Pts Diagnostics
Sekisui Diagnostics
Other Players
Lifescan Ip Holdings, LLC
Ascensia Diabetes Care Holdings Ag
Fluxergy
Prescision Biosensor, Inc.
Acon Laboratories, Inc.
A. Menarini Diagnostics S.R.L
Orasure Technologies, Inc.
Mankind Pharma
Terumo Corporation
Grifols, S.A.
Diasorin S.P.A.
Accubiotech Co., Ltd.
Meridian Bioscience
Biocartis
Genestat Molecular Diagnostics, LLC.
For more information about this report visit https://www.researchandmarkets.com/r/dhsbvb
Attachment
CONTACT: CONTACT: ResearchAndMarkets.com Laura Wood, Senior Press Manager press@researchandmarkets.com For E.S.T Office Hours Call 1-917-300-0470 For U.S./CAN Toll Free Call 1-800-526-8630 For GMT Office Hours Call +353-1-416-8900


 Yahoo Finance
Yahoo Finance 
