Global Clinical Trials Matching Software Market (2022 to 2030) - Size, Share & Trends Analysis Report

Global Clinical Trials Matching Software Market
Dublin, May 30, 2022 (GLOBE NEWSWIRE) -- The "Clinical Trials Matching Software Market Size, Share & Trends Analysis Report by Deployment Mode (Web & Cloud Based, On-premise), by End Use, by Region, and Segment Forecasts, 2022-2030" report has been added to ResearchAndMarkets.com's offering.
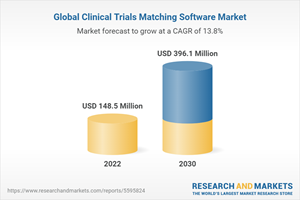
The global clinical trials matching software market size is expected to reach USD 396.1 million by 2030. It is expected to expand at a CAGR of 13.8% from 2022 to 2030.
The significant increase in the number of ongoing clinical trials is likely to drive the market. In addition, the growing adoption of the clinical trial matching software catering to the clinical trials, along with the increased demand for virtual trials and automation in the healthcare sector are some of the key factors contributing to the market growth. The matching software help in effective and fast patient matching with patient-centric approaches.
In clinical trials, patient recruitment or matching can be time-consuming, and finding the right match can be a hurdle. Screening or locating prospective respondents who are qualified, considering all elements of the trials, verifying awareness, and getting informed consent to participate are the factors taken into consideration while recruiting patients.
Enlisting the individuals in accordance with the qualifying requirements is crucial, hence the trial matching technology has been proved to be useful, especially in the COVID-19 scenario.
The software helps not only to find the right match but also saves the R&D-related costs, enabling smoother operations without human intervention. The software providers are introducing new innovative techniques to strengthen their market position. For instance, in February 2022, the CTMA expanded CT-SCOUT technology offering in rheumatology.
Clinical Trials Matching Software Market Report Highlights
Based on deployment mode, the web and cloud based segment dominated the market in terms of revenue in 2021 and it is expected to register the fastest CAGR during the forecast period. The cloud computing models operate with no maintenance or upkeep charges and customers only have to pay for the services that are used. On the other hand, on-premises deployment involves in-house infrastructure, in-house IT support, working capital, and higher integration costs. Hence, web and cloud based models are preferred.
Based on end use, pharmaceuticals and biotechnology companies captured the largest revenue share in 2021 owing to the higher adoption of software during ongoing clinical studies for cost-saving in the R&D activities.
The CROs end-use segment is anticipated to expand at the fastest CAGR of 13.7% during the forecast period. CROs provide the professional assistance, expertise, and execution experience required for clinical trials quickly, without the need for the sponsor to engage such people full-time. CROs are preferred for outsourcing as their services are cost and time-effective.
In 2021, North America led the market in terms of revenue owing to the rising adoption of the clinical trial matching software by the pharma, biotech, and medical companies in the region.
Asia Pacific is anticipated to register the fastest growth rate over the forecast period due to the availability of a large patient pool supporting easy recruitment of patients/candidates.
Key Topics Covered:
Chapter 1 Report Scope and Objectives
Chapter 2 Methodology
Chapter 3 Executive Summary
Chapter 4 Market Variables, Trends & Scope
4.1 Market Lineage Outlook
4.1.1 Parent Market Outlook
4.1.2 Ancillary Market Outlook
4.2 Clinical Trials Matching Software Market Dynamics
4.2.1 Market Driver Analysis
4.2.2 Market Restraint Analysis
4.3 Clinical Trials Matching Software Market: Business Environment Analysis Tools
4.3.1 Porter's Five Forces Analysis
4.3.1.1 Competitive Rivalry
4.3.1.2 Threat of Substitutes
4.3.1.3 Threat of Entrants
4.3.1.4 Bargaining Power of Buyers
4.3.1.5 Bargaining Power of Suppliers
4.3.2 Pestel Analysis
4.3.2.1 Political & Legal
4.3.2.2 Economic & Social
4.3.2.3 Technological
4.4 Penetration & Growth Prospect Mapping
4.5 Impact of COVID-19 on Clinical Trials Matching Software Market
Chapter 5 Clinical Trials Matching Software Market: Deployment Mode Analysis
5.1 Clinical Trials Matching Software Market Deployment Mode Market Share Analysis, 2021 & 2030
5.2 Clinical Trials Matching Software Market Deployment Mode Market: Segment Dashboard:
5.3 Market Size & Forecasts and Trend Analyses, 2017 to 2030 for the Deployment Mode Segment
5.3.1 Web & Cloud Based
5.3.1.1 Web & Cloud Based Market, 2017-2030 (USD Million)
5.3.2 On-Premise
5.3.2.1 On-premise Market, 2017-2030 (USD Million)
Chapter 6 Clinical Trials Matching Software Market: End-use Analysis
6.1 Clinical Trials Matching Software Market End-use Market Share Analysis, 2021 & 2030
6.2 Clinical Trials Matching Software Market End-use Market: Segment Dashboard:
6.3 Market Size & Forecasts and Trend Analyses, 2017 to 2030 for the End-use Segment
6.3.1 Pharmaceutical & Biotechnology Companies
6.3.1.1 Pharmaceutical & biotechnology companies Market, 2017-2030 (USD Million)
6.3.2 CROS
6.3.2.1 CROs Market, 2017-2030 (USD Million)
6.3.3 Medical Device Firms
6.3.3.1 Medical device firms Market, 2017-2030 (USD Million)
Chapter 7 Clinical Trials Matching Software Market: Regional Analysis
Chapter 8 Competitive Analysis
8.1 Recent Developments & Impact Analysis, by Key Market Participants
8.2 Company Profiles
8.2.1 IBM Clinical Development
8.2.1.1 Company overview
8.2.1.2 Financial performance
8.2.1.3 Product benchmarking
8.2.1.4 Strategic initiatives
8.2.2 Antidote Technologies, INC.
8.2.2.1 Company overview
8.2.2.2 Financial performance
8.2.2.3 Product benchmarking
8.2.2.4 Strategic initiatives
8.2.3 Ofni Systems
8.2.3.1 Company overview
8.2.3.2 Financial performance
8.2.3.3 Product benchmarking
8.2.3.4 Strategic initiatives
8.2.4 Sss International Clinical Research
8.2.4.1 Company overview
8.2.4.2 Financial performance
8.2.4.3 Product benchmarking
8.2.4.4 Strategic initiatives
8.2.5 Clario
8.2.5.1 Company overview
8.2.5.2 Financial performance
8.2.5.3 Product benchmarking
8.2.5.4 Strategic initiatives
8.2.6 Advarra
8.2.6.1 Company overview
8.2.6.2 Financial performance
8.2.6.3 Product benchmarking
8.2.6.4 Strategic initiatives
8.2.7 Aris Global
8.2.7.1 Company overview
8.2.7.2 Financial performance
8.2.7.3 Product benchmarking
8.2.7.4 Strategic initiatives
8.2.8 Bsi Business Systems Integration Ag
8.2.8.1 Company overview
8.2.8.2 Financial performance
8.2.8.3 Product benchmarking
8.2.8.4 Strategic initiatives
8.2.9 Teckro
8.2.9.1 Company overview
8.2.9.2 Financial performance
8.2.9.3 Product benchmarking
8.2.9.4 Strategic initiatives
8.2.10 Clinical Trials Mobile Application
8.2.10.1 Company overview
8.2.10.2 Financial performance
8.2.10.3 Product benchmarking
8.2.10.4 Strategic initiatives
For more information about this report visit https://www.researchandmarkets.com/r/w9y8f7
Attachment
CONTACT: CONTACT: ResearchAndMarkets.com Laura Wood, Senior Press Manager press@researchandmarkets.com For E.S.T Office Hours Call 1-917-300-0470 For U.S./CAN Toll Free Call 1-800-526-8630 For GMT Office Hours Call +353-1-416-8900


 Yahoo Finance
Yahoo Finance 
