Global Medical Device Outsourcing Market Report 2022: Increase in Off-Shoring Medical Device Manufacturing to Emerging Economies Driving Growth

Global Medical Device Outsourcing Market
Dublin, Sept. 09, 2022 (GLOBE NEWSWIRE) -- The "Medical Device Outsourcing Market Size, Share & Trends Analysis Report by Application (Cardiology, General & Plastic Surgery, IVD), by Service (Contract Manufacturing, Quality Assurance), by Region, and Segment Forecasts, 2022-2030" report has been added to ResearchAndMarkets.com's offering.
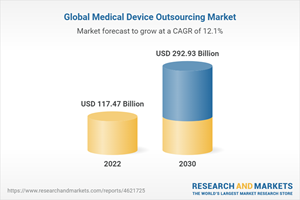
The global medical device outsourcing market size is expected to reach USD 292.93 billion by 2030, registering a CAGR of 12.1% during the forecast period. The rising demand for advanced medical products and a growing trend of offshoring are fueling the growth of this market.
Various initiatives by public organizations promoting testing for COVID-19 are also enhancing market growth. Regulatory bodies are emphasizing the quality of the healthcare devices provided to the public that is becoming burdensome to handle. To meet the regulatory scenario, various services like regulatory consulting are being utilized by the industry players.
For example, the European Medical Device Regulation undertake special spontaneous inspection of outsourced products to test the quality as well as conformity with standards. Contract manufacturers, especially in developed countries like the U.S., EU nations, and Japan, comply with the international standards for a quality management system, which makes them a preferred option as compared with contract manufacturers in India and China.
However, regulatory changes anticipated in developing countries ensure compliance by contract manufacturers. The demand for regulatory compliance is expected to boost the growth of consulting services, such as remediation, compliance, and QMS, thereby contributing to the growth of the market.
Medical Device Outsourcing Market Report Highlights
The contract manufacturing market dominated the global market in 2021 owing to the growing trend of outsourcing by OEMs to third-party manufacturers, especially in emerging countries.
The segment of quality assurance services is projected to exhibit the fastest growth rate over the forecast period due to the increasing trend of outsourcing quality assurance services among emerging countries.
Asia Pacific was the dominant regional market in 2021 owing to lower cost and availability of skilled human resources.
The regional market is estimated to expand further at the second-fastest CAGR during the forecast period.
North America also registered a significant share in 2021 due to well-established infrastructure for manufacturing complex medical devices.
Market Dynamics
Market Drivers
Changing Regulatory Landscape
Increase in Off-Shoring Medical Device Manufacturing to the Emerging Economies
Rising Demand for the Advanced Products
Implementation of the International Standards by the Contract Manufacturers
Rising Price Competition and the Requirement to Reduce Cost
Increasing Complexity With Respect to Product Design and Engineering
Market Restraints
Compliance Issues While Outsourcing
Changing Scenarios Within Developing Countries
Regulatory and Legal Compliance
Contractual Obligation
Industry Challenges
Managing Relationships
Pricing Analysis
Key Topics Covered:
Chapter 1 Methodology and Scope
Chapter 2 Executive Summary
Chapter 3 Medical Device Outsourcing Market: Variables, Trends & Scope
Chapter 4 Medical Device Outsourcing Market: Services Segment Analysis
Chapter 5 Medical Device Outsourcing Market: Application Segment Analysis
Chapter 6 Medical Device Outsourcing Market: Regional Analysis
Chapter 7 Competitive Analysis
Chapter 8 Company Profiles
Chapter 9 KOL Commentary
Chapter 10 Recommendations
Companies Mentioned
SGS SA
Labcorp
Eurofins Scientific
Pace Analytical Services, Inc.
Intertek Group plc
Wuxi Apptec
North American Science Associates, Inc.
Tuv Sud AG
American Preclinical Services
Sterigenics International LLC
Charles River Laboratories International, Inc.
Medical Device Testing Services
Rjr Consulting, Inc
Mandala International.
Freyr
Global Regulatory Partners, LLC
Parexel International Corporation
Emergo
Biotecnika
Accell Clinical Research
Genpact Ltd.
Criterium, Inc.
Pra Health Sciences
Promedica International
Medpace Holdings, Inc.
Icon plc.
Iqvia Holdings Inc.
Integer Holdings Corporation
Tecomet Inc.
Jabil Inc.
Flex Ltd.
Celestica Inc.
Sanmina Corporation.
Plexus Corp.
Phillips Medisize
Cantel Medical Corp.
West Pharmaceutical Services, Inc.
For more information about this report visit https://www.researchandmarkets.com/r/k8vc0v
Attachment
CONTACT: CONTACT: ResearchAndMarkets.com Laura Wood,Senior Press Manager press@researchandmarkets.com For E.S.T Office Hours Call 1-917-300-0470 For U.S./ CAN Toll Free Call 1-800-526-8630 For GMT Office Hours Call +353-1-416-8900


 Yahoo Finance
Yahoo Finance 
