Global Microbiome Therapeutics Market Report 2021-2032: Potential of Microbiome Therapeutics to Address Unmet Needs of Existing Treatment Options

Global Microbiome Therapeutics Market
Dublin, Aug. 03, 2022 (GLOBE NEWSWIRE) -- The "Global Microbiome Therapeutics Market - A Global and Regional Analysis: Focus on Target Therapies, Region (10 Countries), and Competitive Landscape - Analysis and Forecast, 2022-2032" report has been added to ResearchAndMarkets.com's offering.
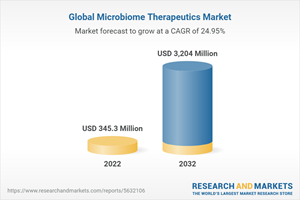
The global microbiome therapeutics market which accounted for $306.3 million in 2021 is expected to reach $3,204 million by 2032, reporting a CAGR of 24.95% during the forecast period 2022-2032.
Microbiome therapeutics have drawn significant attention from the medical community with their potential to address the existing unmet need of various clinical conditions.
The existing microbiome therapeutics market is favored by multiple factors, including the rising geriatric populations coupled with the increasing adoption of inorganic growth strategies by key players in the market.
How Can This Report Add Value to an Organization?
Product/Innovation Strategy: The report provides an exhaustive list of pipeline products which are currently in the development along with their developmental details in various indications. This can help organizations in understanding the research activities, which is the promising indication to pursue or which organization has the potential product to enter first into the market as well it can help the organization to assess where should they place themselves in order to get maximum benefit.
Growth/Marketing Strategy: In addition to pursuing research on exploring the benefits of microbiome therapeutics for new indications, companies are evaluating next-generation-probiotics (NGPs) to address clinical conditions. Looking at the storage issues of microbiome therapeutics, companies are exploring extracellular vesicles as an alternative option. Furthermore, companies are working on increasing consumer awareness about Microbiome therapeutics and its potential benefits of being natural product containing live microorganism derived from human body as well as pursuing synergistic activities to bring an effective microbiome therapeutics for the affected population.
Competitive Strategy: Key players in the global microbiome therapeutics market were analyzed and profiled in the study, who are developing products containing live biotherapeutic products (LBPs), fecal microbiota transplantation (FMT) as well as next-generation-probiotics (NGPs).
Moreover, a detailed competitive benchmarking of the players operating in the global microbiome therapeutics market has been done to help the reader understand how players stand against each other, presenting a clear market landscape. Additionally, comprehensive competitive strategies such as partnerships, agreements, and collaborations will aid the reader in understanding the untapped revenue pockets in the market.
Impact of COVID-19
There were numerous consequences due to the COVID-19 pandemic, especially because of the reduced access to care for other illnesses. As the number of individuals becoming ill from COVID-19 kept increasing and for the protection of healthy individuals from being affected by the disease, all non-urgent healthcare facilities were suspended as per respective government directives.
Even though these measures were necessary, it had mostly negative impact on the Global microbiome therapeutics market. Some the major impacts were; interruption of key clinical trial activities, modification of clinical trial guidelines bringing in additional safety measures, and delay in regulatory agencies' review and approval timeline.
Market Segmentation:
This segmentation highlights value propositions and business models useful for industry leaders and stakeholders. The research also comprises country-level analysis, go-to-market strategies of leading players, and future opportunities, among others, to detail the scope and provide a 360-coverage of the domain.
Target Therapy Area
Disorders related to gastrointestinal (GI) and skin disorders command a major share of the microbiome therapeutics segment, and is expected to continue during the forecast period. Most of the microbiome therapeutics in advanced development stage are indicated to treat C.diff. infection (CDI) and expected to enter the market first. The indications selected were on the basis of research intensity in the specific therapy area.
Region
Asia-Pacific (APAC) is expected to dominate the global microbiome therapeutics market for the forecast period 2022-2032 continuuing with it overall contribution of approximately 40% of the total market size in 2021 followed by Europe with 32.59%. Increasing healthcare expenditure, coupled with government initiatives, are among the leading factors contributing to the growth of the market.
Demand - Drivers and Challenges
Some of the potential drivers identified by BIS Research includes:
Growing Strategic Activities in Microbiome Therapeutics Segment
Potential of Microbiome Therapeutics to Address Unmet Needs of Existing Treatment Options
Microbiome Therapeutic Products as a Safer Alternative to Conventional Drug Treatments
There are some challenges identified for the global microbiome therapeutics market are; Lack of standard regulatory guidelines and safety issues associated with live biotherapeutic products (LBPs).
Key Market Players and Competition Synopsis
The companies that are profiled have been selected based on inputs gathered from primary experts and analyzing company coverage, product portfolio, and market penetration.
The top segment players leading the market include Sanofi S.A and Taisho Pharmaceutical Holding that capture around 88% of the market with Sanofi S.A holding 64.31% while Taisho Pharmaceutical Holdings 24.49% respectively.
Some of the prominent established names in this market are:
4D pharma plc
Seres Therapeutics, Inc.
Microbiotica
Enterome
Enterome Biosciences
Destiny Pharma plc
Taisho Pharmaceutical Holdings
AOBiome Therapeutics, Inc.
Finch Therapeutics Group, Inc.
Rebiotix Inc. (A Subsidiary of Ferring Pharmaceuticals)
MaaT Pharma
Vedanta Biosciences Inc.
OxThera AB
Pendulum Therapeutics
Caelus Health
Quorum Innovations
Sanofi S.A.
DermBiont, Inc.
EnteroBiotix Ltd
YSOPIA Bioscience
Winclove Probiotics
TargEDys
Evelo Biosciences, Inc.
BiomX
Biomica Ltd.
Scioto Biosciences, Inc.
Lactobio A/S
Key Topics Covered:
1 Market Definition
2 Research Scope
3 Research Methodology
4 Market Overview
4.1 Market Size and Future Growth Potential
4.2 Historical Trends
5 Industry Insights
5.1 Regulatory Landscape
5.1.1 Regulatory Requirements for Live Biotherapeutic Products (LBPs)
5.1.2 Quality Check of LBPs
5.1.3 FDA Approved Fecal Microbiota Transplantation (FMT) Quality Check Procedure
5.2 Patent Landscape
5.3 Partnership Landscape
5.4 Government Initiatives
5.5 Impact of COVID-19 on the Global Microbiome Therapeutics Market
5.5.1 Clinical Trial Disruptions and Resumptions
6 Market Dynamics
6.1 Market Drivers
6.1.1 Growing Strategic Activities in Microbiome Therapeutics Segment
6.1.2 Potential of Microbiome Therapeutics to Address Unmet Needs of Existing Treatment Options
6.1.3 Microbiome Therapeutic Products as a Safer Alternative to Conventional Drug Treatments
6.2 Market Restraints
6.2.1 Lack of Uniformed Standardized Regulatory Guidelines
6.2.2 Associated Safety Issues with LBPs
6.2.3 Storage Issues with Live Micro-Organisms
6.2.4 Challenges Associated with Manufacturing of LBPs
6.3 Market Opportunities
6.3.1 Need for Current Good Manufacturing Practice (cGMP) Certified Contract Manufacturing Facilities
6.3.2 Growing Disease Dimension
6.3.3 Entry of Major Players
7 Clinical Trial Landscape
7.1 Microbiome Therapeutics Pipeline Landscape
7.1.1 Competitive Landscaping
7.1.2 Pipeline Analysis
7.1.2.1 By Development Phase
7.1.2.2 By Route of Administration
7.1.2.3 By Type of Microbiome Strategy
7.1.2.4 By Indication
7.1.3 Emerging Microbiome Therapeutics Products
7.2 Probable Potential First Entrants to Global Microbiome Therapeutics Market/ Microbiome Therapeutics Clinical Trial Design
7.2.1 CP-101 (Finch Therapeutics Group, Inc.)
7.2.2 SER-109 (Seres Therapeutics, Inc.)
7.2.3 RBX-2660 (Rebiotix Inc.)
7.2.4 CH-106 (Caelus Health)
7.2.5 LACTIN-V (Osel Inc.)
7.2.6 MaaT013 (MaaT Pharma)
8 Competitive Insights
8.1 Key Strategies and Developments
8.2 Synergistic Activities
8.3 Product Launch and Regulatory Approvals
8.4 Funding and Investment
8.5 Mergers and Acquisitions
8.6 Market Share Analysis
8.7 Growth-Share Analysis (Opportunity Mapping)
8.7.1 By Region
8.7.2 By Company
9 Global Microbiome Therapeutics Market (by Target Therapy Area)
9.1 Overview
9.2 Gastrointestinal and Infectious Diseases
9.3 Skin Disorders
9.4 Cancer Indications
9.5 Other Indications
10 Global Microbiome Therapeutics Market (by Region)
10.1 Overview
10.2 North America
10.2.1 Overview
10.2.2 U.S.
10.2.3 Canada
10.3 Europe
10.3.1 Overview
10.3.2 France
10.3.3 Germany
10.3.4 Sweden
10.3.5 U.K.
10.3.6 Italy
10.3.7 Spain
10.3.8 Rest-of-Europe
10.4 Asia-Pacific (APAC)
10.4.1 Overview
10.4.2 Japan
10.4.3 Australia
10.4.4 China
10.4.5 India
10.4.6 Rest-of-Asia-Pacific
10.5 Rest-of-the-World
10.5.1 Overview
11 Competitive Benchmarking and Company Profiles
For more information about this report visit https://www.researchandmarkets.com/r/lb720m
Attachment
CONTACT: CONTACT: ResearchAndMarkets.com Laura Wood, Senior Press Manager press@researchandmarkets.com For E.S.T Office Hours Call 1-917-300-0470 For U.S./CAN Toll Free Call 1-800-526-8630 For GMT Office Hours Call +353-1-416-8900


 Yahoo Finance
Yahoo Finance 
