Global Narcolepsy Therapeutics Market Report to 2028: Growing Healthcare Expenditures and Increasing Prevalence of Neurological Conditions are Driving Factors

narcolepsy-therapeutics-market-size.jpg
Dublin, Nov. 10, 2022 (GLOBE NEWSWIRE) -- The "Global Narcolepsy Therapeutics Market Size, Share & Industry Trends Analysis Report By Type, By Product (Sodium Oxybate, Selective Serotonin Reuptake Inhibitor, Central Nervous System Stimulants, Tricyclic Antidepressants), By Regional Outlook and Forecast, 2022 - 2028" report has been added to ResearchAndMarkets.com's offering.
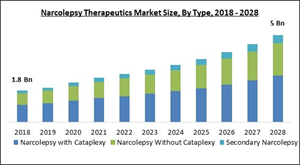
The Global Narcolepsy Therapeutics Market size is expected to reach $5 billion by 2028, rising at a market growth of 10.5% CAGR during the forecast period.
Chronic, incapacitating neurologic disease called narcolepsy is characterized by excessive daytime sleepiness (EDS). Modafinil, armodafinil, and stimulants are only a few of the medications used to treat narcolepsy. Others include sodium oxybate and venlafaxine, which are used to treat the symptoms of sleep paralysis and sleep-related hallucinations (e.g. sodium oxybate).
In general, drugs that boost norepinephrine or dopamine release or prevent their reuptake have wake-promoting effects and can help manage EDS, whereas drugs that block serotonin or norepinephrine reuptake have anti-cataplexy benefits. Both EDS and cataplexy are affected by the modification of either histamine H3 receptors (H3Rs) or -aminobutyric acid B (GABAB) receptors.
The most recent approved medications for EDS related to narcolepsy in the European Union and the USA are solriamfetol, a dopamine and norepinephrine reuptake inhibitor, and pitolisant, an H3R antagonist. Many new drugs are being developed and tested as potential treatments for EDS and cataplexy associated with narcolepsy (THN102), including novel Oxybate formulations of low sodium [FT218] and one-nightly [JZP-258], selective norepinephrine reuptake inhibitors (AXS-12), and modafinil and astroglial connexin inhibitors (THN102).
The mechanisms of action, pharmacokinetics, effectiveness, and safety/tolerability of recently authorized and developing therapies for narcolepsy. A chronic, incapacitating neurologic condition of hypersomnolence called narcolepsy is thought to affect 20-67 persons per 100,000 people across the world. Narcolepsy typically begins in the second decade of life; however, a diagnosis is frequently made much later.
Excessive daytime sleepiness (EDS), while not a specific symptom of narcolepsy, is a feature of the illness that must be present in all individuals to make the diagnosis. Up to 60% of patients have cataplexy, an involuntary decrease of muscle tone while awake that is generally brought on by intense emotions. Restless nights, hypnagogic and hypnopompic hallucinations that happen when the user falls asleep and wakes up, and sleep paralysis are other symptoms.
COVID-19 Impact Analysis
During the pandemic, COVID-19 had a detrimental effect on narcolepsy medications. The biggest problem is that patients with narcolepsy can stop taking their medication because they are unable to get their prescriptions filled because of transportation limitations, home quarantines, and hospital cross-infection fears. Additionally, because there is no effective treatment for COVID-19, manufacturers and companies are concentrating more on producing the pharmaceuticals and tools that are required for treating COVID-19.
Market Growth Factors
Growing Healthcare Expenditures
One of the main factors boosting the market's rise is the rising number and size of investments made in the healthcare sector by governments of various countries. It is anticipated that more developing nations is expected to have greater access to high-quality healthcare as a result of their quick economic development and growing healthcare costs. This is viewed as a positive indicator of expansion for the narcolepsy therapeutics industry. The need for various treatment and apparatus has surged in these nations due to the rising insomnia problems.
Increasing Prevalence Of Neurological Conditions
Neurologists treat a wide variety of sleep problems, including narcolepsy, sleep apnea, restless legs syndrome, insomnia, and sleep apnea. Endogenous irregularities are brought on by several illnesses, which interfere with the regular sleep-wake cycle. In the upcoming years, neurological sleep-related diseases are predicted to rise as a significant section of the population ages. The National Institute of Health (NIH) estimates that 20 million Americans occasionally have sleep issues, whereas 40 million Americans suffer from neurological illnesses.
Market Restraining Factors
High Cost Of Narcolepsy Diagnosis & Treatment
One of the main factors holding back the narcolepsy market is the high cost of narcolepsy diagnosis and treatment. Additionally, the term "sleeping problem" refers to a broad range of illnesses with serious negative effects on the health of the individual and huge societal costs. Furthermore, in 2019 the smooth operation of the narcolepsy market is disrupted by the high cost of therapy. Daily functioning is hampered by narcolepsy, which is also connected with a significant financial and medical burden - many patients are fully disabled.
Key Topics Covered:
Chapter 1. Market Scope & Methodology
Chapter 2. Market Overview
2.1 Introduction
2.1.1 Overview
2.1.1.1 Market composition & scenarios
2.2 Key Factors Impacting the Market
2.2.1 Market Drivers
2.2.2 Market Restraints
Chapter 3. Global Narcolepsy Therapeutics Market by Type
3.1 Global Narcolepsy with Cataplexy Market by Region
3.2 Global Narcolepsy Without Cataplexy Market by Region
3.3 Global Secondary Narcolepsy Market by Region
Chapter 4. Global Narcolepsy Therapeutics Market by Product
4.1 Global Sodium Oxybate Market by Region
4.2 Global Selective Serotonin Reuptake Inhibitor Market by Region
4.3 Global Central Nervous System Stimulants Market by Region
4.4 Global Tricyclic Antidepressants Market by Region
4.5 Global Others Market by Region
Chapter 5. Global Narcolepsy Therapeutics Market by Region
Chapter 6. Company Profiles
6.1 Jazz Pharmaceuticals plc
6.1.1 Company Overview
6.1.2 Financial Analysis
6.1.3 Regional Analysis
6.1.4 Research & Development Expenses
6.1.5 Recent strategies and developments:
6.1.5.1 Partnerships, Collaborations, and Agreements:
6.1.5.2 Approvals and Trials:
6.2 Shionogi & Co., Ltd.
6.2.1 Company Overview
6.2.2 Financial Analysis
6.2.3 Research & Development Expenses
6.3 Teva Pharmaceutical Industries Ltd.
6.3.1 Company Overview
6.3.2 Financial Analysis
6.3.3 Regional Analysis
6.3.4 Research & Development Expenses
6.4 Pfizer, Inc. (Arena Pharmaceuticals, Inc.)
6.4.1 Company Overview
6.4.2 Financial Analysis
6.4.3 Regional & Segmental Analysis
6.4.4 Research & Development Expense
6.5 Bioprojet
6.5.1 Company Overview
6.5.2 Recent strategies and developments:
6.5.2.1 Partnerships, Collaborations, and Agreements:
6.6 Takeda Pharmaceutical Company Limited
6.6.1 Company Overview
6.6.2 Financial Analysis
6.6.3 Regional Analysis
6.6.4 Research & Development Expense
6.7 Ligand Pharmaceuticals, Inc.
6.7.1 Company Overview
6.7.2 Financial Analysis
6.7.3 Research & Development Expenses
6.8 Graymark Healthcare, Inc.
6.8.1 Company Overview
For more information about this report visit https://www.researchandmarkets.com/r/maubkq
Attachment
CONTACT: CONTACT: ResearchAndMarkets.com Laura Wood,Senior Press Manager press@researchandmarkets.com For E.S.T Office Hours Call 1-917-300-0470 For U.S./ CAN Toll Free Call 1-800-526-8630 For GMT Office Hours Call +353-1-416-8900


 Yahoo Finance
Yahoo Finance 
