Global Pediatric Vaccines Market Analysis Report 2022-2027: Increased Demand from Emerging Markets, Growing Investments and R&D, Collaboration Agreements, Product Approvals and Launches

Global Pediatric Vaccines Market
Dublin, Nov. 17, 2022 (GLOBE NEWSWIRE) -- The "Pediatric Vaccines: Global Markets" report has been added to ResearchAndMarkets.com's offering.
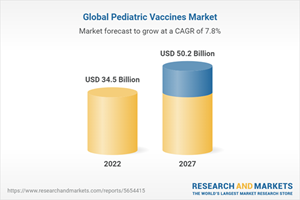
This report highlights the current and future market potential of pediatric vaccines and a detailed analysis of the market's drivers, challenges and opportunities. The report also covers market projections through 2027 and estimates market shares for key players, as well as pediatric vaccines by type and by application. The report also discusses the impact of the COVID-19 pandemic on this market.
Vaccines are one of the most significant advances in global health and are the most cost-effective strategy to reduce child mortality. Vaccines help to prevent 2 million to 3 million deaths each year. Yet, each year millions of children are left out of vaccine distribution, mostly in less developed regions.
The COVID-19 pandemic also affected the pediatric immunization schedules. As per WHO, the vaccine coverage dipped for major vaccines such as those for diphtheria, tetanus and pertussis, measles, mumps and varicella, BCG and hepatitis. The dip in coverage led to outbreaks of vaccine-preventable diseases in some regions. As a result, organizations such as Gavi, UNICEF and WHO have started taking measures to increase the pediatric vaccine coverage.
Pneumococcal, meningococcal, combination vaccine (hexavalent vaccine) and COVID-19 have been approved. In 2021, vaccines such as Comirnaty from Pfizer/BioNTech, Spikevax from Moderna, CoronaVac from Sinovac, vaccines from the China National Pharmaceutical Group (Sinopharm), ZyCoV-D from Zydus Cadila (India) and Covaxin from Bharat Biotech (India) have been approved for the pediatric population.
Other drivers contributing to the market include government support, growing investments and R&D budgets, collaboration agreements, and the increasing incidence and burden of diseases.
In this report, the market is segmented based on technology, disease type and region. The North American market currently holds the highest market share.
Company profiles of major players within the industry, including Pfizer, Abbott Laboratories, Merck & Co., GlaxoSmithKline, Sanofi SA and AstraZeneca
Report Includes
Analyses of the global market trends, with historic market revenue data for 2020 and 2021, estimates for 2022, and projections of compound annual growth rates (CAGRs) through 2027
Estimation of the actual market size for pediatric vaccines in USD million values, market forecast and corresponding market share analysis by technology, disease type (application), and geographic region
Highlights of the key growth driving factors and constraints that will shape the market for pediatric vaccines as the basis for projecting demand over the forecast period (2022-2027)
In-depth information (facts and figures) concerning major market dynamics (DROs), technology advancements, regulatory aspects, and other macroeconomics factors affecting the marketplace
Highlights of the COVID-19 impact on the global pediatric vaccines market, with pandemic implications on demand/supply of vaccines across the world, and govt. strategic decisions to boost the marketplace
Emphasis on the new developments in the pediatric vaccines market, and underlying clinical trials for all phases and list of vaccines
Identification of the major stakeholders and analysis of the competitive landscape based on recent developments, financial performance, and segmental revenues
Key Topics Covered:
Chapter 1 Introduction
Chapter 2 Summary and Highlights
Chapter 3 Technology Background
3.1 Introduction
3.1.1 History of Vaccine Development
3.1.2 Types of Vaccines
3.1.3 Attenuated (Live) Vaccines
3.1.4 Inactivated (Killed) Vaccines
3.1.5 Toxoid Vaccines
3.1.6 Subunit Vaccines
3.1.7 Conjugate Vaccines
3.1.8 Recombinant Vector Vaccines
3.1.9 Dna Vaccines
3.1.10 Rna Vaccines
3.1.11 Childhood Vaccination Policies
3.1.12 Pediatric Immunization
3.2 Regulatory Aspects
3.2.1 New Product Approvals
Chapter 4 Market Dynamics
4.1 Factors Affecting the Pediatric Vaccine Market
4.2 Market Drivers
4.2.1 Increasing Awareness and Government Support
4.2.2 Growing Investments and R&D
4.2.3 Product Approvals and Launches
4.2.4 Collaboration Agreements
4.2.5 Increasing Burden of Diseases
4.3 Market Restraints
4.3.1 Investment and Pricing
4.4 Market Opportunities
4.4.1 Increased Demand from Emerging Markets
Chapter 5 Impact of Covid-19 Pandemic
5.1 Introduction
5.2 Epidemiology
5.3 Impact of Covid-19 on the Pediatric Vaccine Market
Chapter 6 Pediatric Vaccine Market, by Technology
6.1 Global Market for Pediatric Vaccines, by Technology
6.2 Live Attenuated Vaccines
6.3 Inactivated Vaccines
6.4 Subunit, Recombinant, Polysaccharide and Conjugate Vaccines
6.5 Toxoid Vaccines
6.6 Other/Combination Vaccines
Chapter 7 Pediatric Vaccine Market, by Disease Type
7.1 Pneumococcal
7.2 Diphtheria, Tetanus and Pertussis
7.3 MMRV
7.4 HPV
7.5 Influenza
7.6 Other Diseases
Chapter 8 Pediatric Vaccine Market, by Region
8.1 Global Market for Pediatric Vaccines, by Region
8.2 North America
8.2.1 United States
8.2.2 Canada
8.3 Europe
8.3.1 Germany
8.3.2 United Kingdom
8.3.3 France
8.3.4 Italy
8.3.5 Spain
8.3.6 Rest of Europe
8.4 Asia-Pacific
8.4.1 China
8.4.2 Japan
8.4.3 India
8.4.4 Rest of Asia-Pacific
8.5 Rest of the World
Chapter 9 New Developments
9.1 Phase I Clinical Trials
9.2 Phase II Clinical Trials
9.3 Phase III Clinical Trials
Chapter 10 Competitive Landscape
10.1 Market Shares of Pediatric Vaccine Companies
10.2 Strategic Initiatives
Chapter 11 Company Profiles
Abbott Laboratories
Astrazeneca plc
Bharat Biotech International Ltd.
Csl Seqirus UK Ltd.
Emergent Biosolutions Inc.
GlaxoSmithKline
Merck & Co, Inc.
Panacea Biotec Ltd.
Pfizer Inc.
Sanofi S.A.
Serum Institute of India Ltd.
Sinovac Biotech Ltd
For more information about this report visit https://www.researchandmarkets.com/r/db5mxn
Attachment
CONTACT: CONTACT: ResearchAndMarkets.com Laura Wood,Senior Press Manager press@researchandmarkets.com For E.S.T Office Hours Call 1-917-300-0470 For U.S./ CAN Toll Free Call 1-800-526-8630 For GMT Office Hours Call +353-1-416-8900


 Yahoo Finance
Yahoo Finance 
