Incyte (INCY) Receives FDA Approval for Skin Cancer Treatment
Incyte INCY announced that the FDA granted accelerated approval to biologics license application (BLA) for the intravenous PD-1 inhibitor retifanlimab-dlwr. The BLA was seeking approval for the treatment of adults with metastatic or recurrent locally advanced merkel cell carcinoma (MCC).
The drug has been approved under the brand name Zynyz.
The accelerated approval was granted based on the tumor’s response rate and duration of response. Continued approval of Zynyz for this indication may be contingent on verification and description of clinical benefit in confirmatory trials.
MCC, a rare and aggressive type of skin cancer, most often appears as a single, painless, reddish-purple skin nodule on the head, neck and arms in skin exposed to sunlight.
The FDA approval was based on data from the POD1UM-201 trial, an open-label, multiregional, single-arm study of Zynyz in adults with metastatic or recurrent locally advanced MCC who had not received prior systemic therapy for their advanced disease.
Zynyz monotherapy resulted in an objective response rate of 52% among chemotherapy-naïve patients. A complete response was seen in 12 patients (18%) and 22 patients (34%) showed partial response.
The POD1UM program for retifanlimab includes POD1UM-201 and several other studies for patients with solid tumors, including registration-directed POD1UM trials evaluating retifanlimab as a monotherapy for patients with microsatellite instability, high endometrial cancer and squamous cell carcinoma of the anal canal (SCAC) and in combination with platinum-based chemotherapy for patients with non-small cell lung cancer and SCAC.
In July 2021, the FDA issued a complete response letter for the BLA of retifanlimab for the treatment of SCAC. In October 2021, Incyte withdrew the MAA seeking approval of retifanlimab for SCAC.
The phase III POD1UM-304 study is evaluating retifanlimab in combination with platinum-based chemotherapy as a first-line treatment for patients with non-small cell lung cancer.
Incyte licensed retifanlimab from MacroGenics, Inc. in 2017.
Shares of Incyte have lost 6.3% in the past year compared with the industry’s decline of 15.6%.
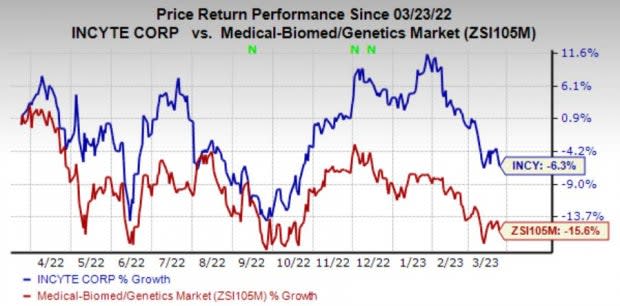
Image Source: Zacks Investment Research
Approval of additional drugs will add an incremental stream of revenues to the top line and reduce the company’s dependence on Jakafi. Incyte has a collaboration agreement with Swiss pharma giant Novartis NVS for Jakafi.
Jakafi’s (a first-in-class JAK1/JAK2 inhibitor approved for polycythemiavera, myelofibrosis and refractory acute graft-versus-host disease). Jakafi is marketed by Incyte in the United States and by Novartis as Jakavi outside the country.
Incyte currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Some better-ranked stocks in the overall healthcare sector are Novo Nordisk NVO and Ligand Therapeutics LGND, both sporting a Zacks Rank #1 at present.
In the past 30 days, estimates for Novo Nordisk’s 2023 earnings per share have risen from $4.20 to $4.43. Estimates for 2024 have increased by 29 cents to $5.19.
Ligand’s earnings per share estimates for 2023 increased to $4.32 from $3.30 in the past 30 days. LGND beat earnings estimates in one of the last four reported quarters.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Novartis AG (NVS) : Free Stock Analysis Report
Novo Nordisk A/S (NVO) : Free Stock Analysis Report
Incyte Corporation (INCY) : Free Stock Analysis Report
Ligand Pharmaceuticals Incorporated (LGND) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 