New ‘iron lung’ to help NHS patients breathe could be ready this year
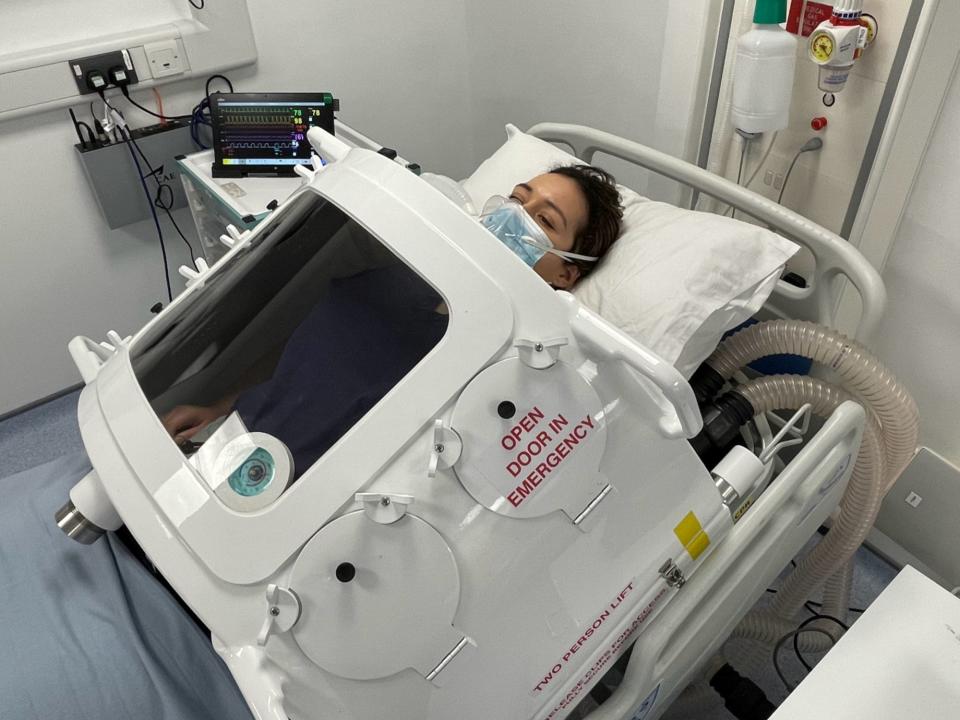
The exovent uses traditional ‘iron lung’ technology to help patients breathe without being sedated
( )A 21st century version of the old “iron lung” technology, which was used to help thousands of patients with polio to breathe, could be available on NHS wards as early as this year.
A team of doctors, scientists and engineers are about to make a bid to the UK’s medical devices regulator to get approval for the use of the exovent device, which they say could prevent some patients needing to be sedated and put on invasive ventilators with tubes down their windpipe.
The exovent uses the same principle as the old iron lungs by creating a negative pressure vacuum around the patient which gently forces air to be sucked into the lungs. It can be used to support patients to breathe or it can take over their breathing completely.
Because the device fits over the patient on a hospital bed, they don’t need to be sedated and can remain awake, eating and drinking and talking.
The team behind the project, who have set up a charity (called Exovent) to help develop their idea, began work on it last year in response to fears the UK and other countries could run out of ventilators because of the demand from patients sick with the coronavirus.
Ian Joesbury, chief executive of Exovent and a former aerospace mechanical engineer, told The Independent: “There are enormous benefits, not just to the NHS here in the UK, but globally. Every 40 seconds a child dies of pneumonia and we are developing a lower cost model of exovent which could be used to treat these patients because you don’t need an intensive care unit or an anaesthetist.
“The exovent could reduce the frequency of patients being ventilated where they are knocked out and a tube put down their throat and not knowing whether they are going to wake up or not. With exovent patients can stay awake throughout the process.
“There are lots of patients with conditions like chronic obstructive pulmonary disease and pneumonia who need respiratory support and this is a much kinder way to deliver that support and it reduces the risk of lung damage which exists with mechanical ventilation.
“It is incredibly gentle.”
The exovent has yet to be investigated through a full clinical trial but it has been tested by several NHS hospitals including the University Hospitals Birmingham Trust which carried out tests demonstrating it worked.
The group now plan to submit a bid to the Medicines and Healthcare products Regulatory Agency and expect it could be given the go ahead within a matter of months.
“We are incredibly confident and we think it could be in use as soon as August, September or October this year,” Mr Joesbury said.
The University Hospital Southampton, University College Hospital London and the Queen Elizabeth Hospital King’s Lynn have all expressed an interest in using the device.
According to a paper in Anaesthesia, a journal of the Association of Anaesthetists, six volunteers, including three senior anaesthetists, have been used to test prototypes built by Marshall Aerospace and Defence Group.
All of the volunteers were tested facing up and slightly tilted, while three were also tested in the prone, or facing down, position. Various pressure settings were used and lung performance readings taken.
These showed that the exovent was able to deliver an increased lung expansion to people breathing spontaneously, and powerful ventilation to take over people’s breathing entirely, using only moderate negative pressures.
It would need less nursing care to look after patients compared with ventilated patients, who are fully dependent on machines and nurses to keep them alive. It could be used in general wards and potentially even in a patients’ home.
During the coronavirus pandemic, hospitals have had to stretch nurse staffing to dangerous levels because of the demand for critical care and oxygen from patients. While some patients have needed to be ventilated others have been treated with masks to force air into their lungs.
Mr Joesbury said the exovent could become an additional tool for clinicians to help patients avoid being ventilated as a last resort. It can also reduce the demand on oxygen systems, which has seen some hospitals forced to declare major incidents and divert patients because of fears the supplies would be cut off.
The patient can be observed through a window and portholes that seal around the arms allow nurses to access the patients body if they need to.
According to the tests, nurses reported the chamber could be positioned and removed quickly by two people and they could monitor and look after patients safely despite the reduced access to.
The report added: “The volunteers all found the chamber comfortable, and in particular reported that the neck and hip seals were soft and easy to adjust, and that they could voluntarily breach them to stretch their arms or touch their faces without this significantly affecting the stability of the chamber pressure.
“Importantly, all the test subjects felt ‘in control’ knowing that they could immediately release the vacuum by opening a wide gap under one of the seals if they wished. When the ventilation mode was used, the subjects all allowed the exovent to take over their breathing without feeling any desire to ‘fight it’ or that it felt unnatural or uncomfortable, and described the sensation as relaxing; one participant fell asleep within minutes.”
It is estimated the UK version of the exovent will cost approximately £8,000, considerably cheaper than existing devices, which cost around £15,000 for CPAP masks and over £30,000 for intensive care ventilators.
A lower-cost global version could be produced for less than £500.

 Yahoo Finance
Yahoo Finance 