Medtronic (MDT) Progresses in Neuromodulation on New FDA Nod
Medtronic’s MDT growing Neuromodulation arm received a major breakthrough following the FDA approval and consequent first U.S. implants of its SenSight Directional Lead System. SenSight is an unique kind of Deep Brain Stimulation (DBS) directional lead that for the first time combines the benefits of directionality with the power of sensing.
SenSight claims to deliver precise, patient-specific DBS therapy for the treatment of some symptoms associated with diseases like Parkinson’s, dystonia and essential tremor as well as medically refractory epilepsy.
DBS and SenSight
In DBS therapy, a small pacemaker-like device sends electrical signals through very thin wires, known as leads to a targeted area in the brain related to symptoms of certain neurological disorders. Post surgery, in another few weeks, a neurologist will wirelessly adjust the neurostimulator setting, which is called programming. The programming aims at controlling symptoms while minimizing potential side effects.
Medtronic’s DBS allows for more precise stimulation and streamline the surgical procedure. At the same time, it also captures objective data for more efficient, informed programming.
According to the company, SenSight is the first directional, sensing-enabled lead designed to enhance the detection of local field potentials (LFPs). LFPs are brain signals that correlate with the severity of Parkinson's disease symptoms and are 1 million times smaller than DBS stimulation pulses.
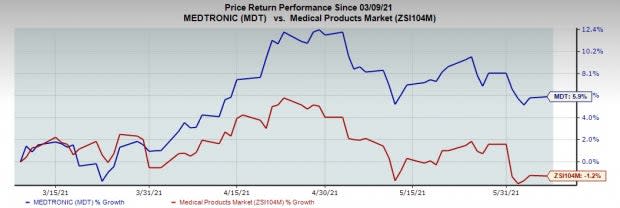
Image Source: Zacks Investment Research
When paired with the Percept PC neurostimulator device, SenSight expands on BrainSense technology, which helps to capture and record enhanced, directional LFP information from the implanted lead.
Superiority of SenSight over Other DBS at a Glance
Traditionally, a single DBS system was unable to provide both sensing capability and directional leads. Accordingly, physicians had to pick only one of the technologies foregoing the other, based on the predicted needs of each patient.
The latest SenSight directional lead combines the benefits of both technologies as it can work in combination with a pulse generator capable of brain sensing. Also, it has ability to continuously record brain activity while affected patients perform their daily activities. Per the press release, this is a powerful research tool that is rapidly improving the understanding of these brain circuitry disorders that affect the lives of so many people.
The first U.S. implant of SenSightwas made at the University of Florida in early June. The full U.S. launch is expected to take place following this latest announcement.
Industry Prospects
Per a report by Research And Markets, the global DBS market is expected to witness a CAGR of 9% between 2019 and 2024. Factors like the frequent occurrence of motor and neurological disorders along with the growing acceptance of minimally invasive surgeries are expected to drive the market.
Given the market potential, the approval has been timed well.
Medtronic’s Recent Strides in Neuromodulation Space
In Neuroscience portfolio, in March 2021, Medtronic received CE Mark for SenSight directional lead system for DBS therapy as treatment of symptoms associated with movement disorders and epilepsy.
In January, the company announced the first enrollment in ADAPT-PD (Adaptive DBS Algorithm for Personalized Therapy in Parkinson's Disease), its trial evaluating the safety and efficacy of adaptive deep brain stimulation (aDBS) in patients with Parkinson's Disease (PD).
Currently, Medtronic awaits the launch of its Vanta recharge-free spinal cord stimulator in the first half of fiscal 2022. It is also on track to submit the ECAPS device to the FDA later in the calendar year 2021.
Share Price Performance
The stock has outperformed its industry over the past three months. It has risen 5.9% against the industry’s 1.2% drop.
Zacks Rank and Key Picks
Currently, the company carries a Zacks Rank #3 (Hold).
A few better-ranked stocks from the broader medical space are Envista Holdings Corporation NVST, Inogen, Inc INGN and IDEXX Laboratories, Inc. IDXX, each carrying a Zacks Rank #2 (Buy). You can see the complete list of Zacks #1 Rank (Strong Buy) stocks here.
Envista Holdings has an estimated long-term earnings growth rate of 26%.
Inogen has an estimated long-term earnings growth rate of 33%.
IDEXX Laboratories has a projected long-term earnings growth rate of 20%.
More Stock News: This Is Bigger than the iPhone!
It could become the mother of all technological revolutions. Apple sold a mere 1 billion iPhones in 10 years but a new breakthrough is expected to generate more than 77 billion devices by 2025, creating a $1.3 trillion market.
Zacks has just released a Special Report that spotlights this fast-emerging phenomenon and 4 tickers for taking advantage of it. If you don't buy now, you may kick yourself in 2022.
Click here for the 4 trades >>
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Medtronic PLC (MDT) : Free Stock Analysis Report
IDEXX Laboratories, Inc. (IDXX) : Free Stock Analysis Report
Inogen, Inc (INGN) : Free Stock Analysis Report
Envista Holdings Corporation (NVST) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 