Middle East Molecular Diagnostics Market Report 2022: Increasing Investments in R&D - Market to Reach a Value of $1,017.7 Million by 2031 from $493.1 Million in 2020

Middle Eastern Molecular Diagnostics Market
Dublin, June 22, 2022 (GLOBE NEWSWIRE) -- The "Middle East Molecular Diagnostics Market - Country Analysis: Focus on Product, Testing Location, Technology, Application, End User, and Country Data - Analysis and Forecast, 2021-2031" report has been added to ResearchAndMarkets.com's offering.
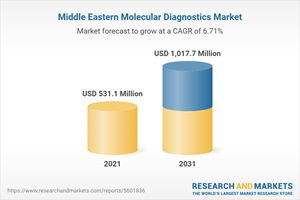
The Middle East molecular diagnostics market is projected to reach $1,017.7 million by 2031 from $493.1 million in 2020, at a CAGR of 6.72% during the forecast period 2021-2031
The growth in the Middle East molecular diagnostics market is expected to be driven by factors such as the increasing prevalence of infectious diseases and various types of cancer in the Middle East, rising awareness of personalized medicine and its acceptance, coupled with the significant number of funding for executing R&D in Middle East region.
Market Lifecycle Stage
Increasing investments in the R&D for molecular diagnostics is one of the major opportunities in the Middle East molecular diagnostics market.
Diagnostics is a well-developed market with an integral part of medical decision-making -aid in acquiring information for a wide range of treatment dissemination, accentuating their criticality in healthcare. Molecular diagnostics has a critical role in the precision medicine approach, as it ensures the safe and effective application of targeted therapeutics. Most of the players in the Middle East molecular diagnostics market offer products encompassing the kits and reagents, instruments, and software categories.
Several diagnostics and pharmaceutical companies are working collaboratively to develop next-generation sequencing (NGS), polymerase chain reaction (PCR), immunohistochemistry (IHC), in-situ hybridization (ISH), microarray, nucleic acid amplification-, or mass spectrometry-based molecular diagnostics for the applications in several disease indications in order to enable efficient diagnosis, treatment selection, dosage selection, and treatment monitoring.
Market Segmentation
Product (Systems, Kits and Consumables, Software and Other Products)
The Middle East molecular diagnostics market in the products segment is expected to be dominated by the kits and consumables segment. This is due to an increasing number of molecular diagnostics kit providers offering kits and services to their end users.
Testing Location (Laboratory Testing, Point-of-Care Testing)
The Middle East molecular diagnostics market is dominated by the laboratory testing segment owing to an increasing number of molecular diagnostic laboratories in the Middle East region involved in undertaking various routine testing for different chronic and acute diseases.
Technology (Polymerase Chain Reaction (PCR), Next-Generation Sequencing (NGS), Isothermal Nucleic Acid Amplification Technology (INAAT), Microarray, In-Situ Hybridization (ISH), Immunohistochemistry (IHC), Other Technologies)
The polymerase chain reaction segment dominates the Middle East molecular diagnostics market due to the increasing number of PCR testing being carried out in the Middle East laboratories for diagnosis.
Application (Core Molecular Diagnostics, Reproductive Genetics, Companion Diagnostics, Liquid Biopsy, Others)
The core molecular diagnostic segment dominates the Middle East molecular diagnostics market due to the rising number of molecular testing in laboratories to cure diseases. Core molecular diagnostics applications include a large number of inclusive applications that cater to the major portion of the Middle East molecular diagnostics market.
End User (Hospitals, Diagnostic Centres, Outpatient Clinics/General Practitioners, Research Laboratories, Others)
The hospitals segment dominates the Middle East molecular diagnostics market as hospitals, particularly in Middle East countries have incorporated extensive molecular diagnostics portfolios to provide superior care to patients suffering from diseases.
Country (Kingdom of Saudi Arabia (K.S.A.), Israel, United Arab Emirates (U.A.E.), Egypt, Iran, Qatar, Other Countries)
K.S.A. generated the highest revenue of $132.5 million in 2020. The Kingdom of Saudi Arabia (K.S.A.) has a huge population base suffering from various diseases and rising healthcare costs in the region. Medical research in the Kingdom of Saudi Arabia (K.S.A.) has gained momentum since 2013 and is heavily being invested in by the local government. The government is extensively supporting research organizations with projects aimed at improving the understanding and treatment of diseases affecting the Saudi population.
Recent Developments in Middle East Molecular Diagnostics Market
In January 2022, Abbott showcased its life-changing diagnostic tools and medical devices product portfolio for point-of-care testing (POCT) that helped COVID-19, diabetes, and cardiovascular disease patients at MEDLAB Middle East.
In June 2021, biomeruix launched EPISEQ SARS-COV-2 to identify SARS-CoV-2 variants using samples from COVID-19 positive patients.
In 2021, the BD. company declared the approval of the BD Veritor At-Home COVID-19 Test for the detection of SARS-CoV-2 with definitive digital results at home.
In 2020, Bio-Rad Laboratories, Inc. partnered with Seegene, Inc., a Middle East leader in multiplex molecular diagnostics, for the commercialization and clinical development of molecular diagnostic products.
Market Dynamics
Market Drivers
Increasing Prevalence of Infectious Diseases and Various Types of Cancer in the Middle East
Increase in Awareness and Acceptance of Personalized Medicines in the Middle East
Significant External Funding for Executing Research and Development Exercises
Market Restraints
Uncertain Reimbursement Scenario
Lack of High-Complexity Testing Centers in the Middle East
Market Opportunities
Massive Scope for Adoption of Molecular Diagnostics
Rise of Next-Generation Ultrasensitive Molecular Diagnostics
Novel Revenue Streams
COVID-19 Impact on Middle East Molecular Diagnostics Market
Impact on Market Size
Impact on Molecular Diagnostic Practices
Key Market Players and Competition Synopsis
Major manufacturers of the Middle East molecular diagnostic market, along with the service providers, are actively involved in undertaking significant business strategies to translate success in research and development into the commercial clinical setting.
Some of the prominent names established in this market are:
Abbott
Agilent Technologies, Inc.
bioMerieux SA
BD
Babirus Medical Equipment LLC
Bio-Rad Laboratories, Inc.
Danaher
F. Hoffmann-La Roche Ltd.
Guardant Health
Illumina, Inc.
QIAGEN N.V.
Thermo Fisher Scientific Inc.
For more information about this report visit https://www.researchandmarkets.com/r/dj366s
Attachment
CONTACT: CONTACT: ResearchAndMarkets.com Laura Wood, Senior Press Manager press@researchandmarkets.com For E.S.T Office Hours Call 1-917-300-0470 For U.S./CAN Toll Free Call 1-800-526-8630 For GMT Office Hours Call +353-1-416-8900


 Yahoo Finance
Yahoo Finance 
