Novartis Announces Positive Data on Breast Cancer Drug Kisqali
Novartis NVS announced positive data on the breast cancer drug Kisqali (ribociclib) from the late-stage NATALEE study at the 2023 American Society of Clinical Oncology (ASCO) Annual Meeting.
NATALEE is a global phase III multi-center, randomized, open-label trial to evaluate the efficacy and safety of Kisqali with endocrine therapy (ET) as adjuvant treatment compared with ET alone in patients with hormone receptor-positive/human epidermal growth factor receptor 2-negative (HR+/HER2-) early breast cancer.
Approximately 5,101 adult patients with HR+/HER2- EBC across 20 countries were randomized for the study.
The primary endpoint of NATALEE is meaningful invasive disease-free survival (iDFS) as defined by the Standardized Definitions for Efficacy End Points (STEEP) criteria.
Data from the study showed that Kisqali plus ET compared with ET alone, lowered the risk of cancer recurrence by 25.2% in patients with stage II and III HR+/HER2- early breast cancer, along with a consistent, clinically iDFS benefit across key pre-specified subgroups.
Data across all secondary efficacy endpoints were also consistent, including distant disease-free survival (DDFS) (26% risk reduction) and recurrence-free survival (RFS) (28% risk reduction), with a trend for improvement in overall survival (OS).
The safety profile of Kisqali at 400 mg was favorable. Novartis plans to submit these phase III data to regulatory authorities in the US and Europe before end-of-year.
Kisqali is approved in the United States for the treatment of adult patients with HR+/HER2- advanced or metastatic breast cancer in combination with an aromatase inhibitor as initial ET or fulvestrant as initial ET or following disease progression on ET in post-menopausal women.
Novartis is further evaluating Kisqali for various stages of breast cancer. It has collaborated with SOLTI, which is leading the HARMONIA study to evaluate whether Kisqali changes tumor biology to enable a better response to ET compared with Ibrance (palbociclib) in patients with HR+/HER2-, HER2-enriched subtype MBC. The company has also partnered Akershus University Hospital in Norway on the NEOLETRIB trial, a neoadjuvant phase II study evaluating Kisqali in HR+/HER2- EBC to discover the potentially unique underlying mechanism of action.
Shares of Novartis have gained 9.6% in the year so far compared against the industry’s 0.2% decline.
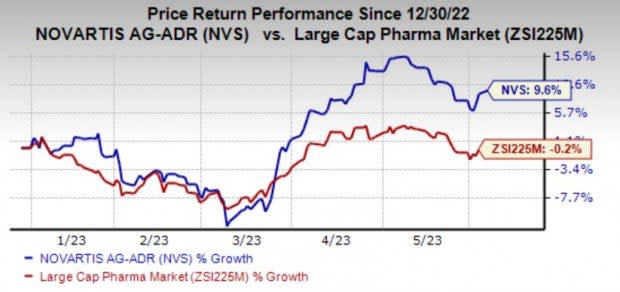
Image Source: Zacks Investment Research
In March, Novartis announced positive top-line results from the NATALEE study on Kisqali. The Independent Data Monitoring Committee recommended that the study be stopped early as the primary endpoint of iDFS was met.
The positive top-line results from NATALEE support Novartis’ efforts to expand the benefits of Kisqali to patients with earlier stages of breast cancer. Kisqali's sales gained 81% in the first quarter to $415 million and should gain further momentum.
Novartis’ performance in the first quarter was better than expected, as earnings and sales beat estimates and guidance was raised. This boosted investors’ sentiment and increased its share price.
While the older drugs face generic competition, the continued strong performance of Entresto, Pluvicto, Kesimpta and Kisqali fueled growth and should maintain momentum. Pluvicto and Scemblix saw very strong launches and recorded solid sales. Demand for Pluvicto continues to exceed supply in the United States. The Leqvio launch continues to progress well.
However, the performance of Novartis’ other growth driver, Cosentyx, was pretty ordinary in the first quarter, as sales growth across key geographies was impacted by revenue deduction adjustments in the United States.
Novartis currently carries a Zacks Rank #2 (Buy). Other top-ranked stocks in the healthcare sector are Ligand Pharmaceuticals LGND and Novo Nordisk NVO, both sporting a Zacks Rank #1 (Strong Buy). You can see the complete list of today’s Zacks #1 Rank stocks here.
Over the past 30 days, earnings estimates for LGND have increased by $1.09 per share to $5.25. LGND topped earnings estimates in two of the last four quarters and missed in the remaining two, the average surprise being 21.50%.
Over the past 30 days, estimates for NVO’s 2023 earnings per share have risen by 23 cents to $5.07. Novo Nordisk topped earnings estimates in three of the last four quarters and missed the same in the remaining one, the four-quarter earnings surprise being 0.35%, on average.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Novartis AG (NVS) : Free Stock Analysis Report
Novo Nordisk A/S (NVO) : Free Stock Analysis Report
Ligand Pharmaceuticals Incorporated (LGND) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 