Outlook on the RNA Based Therapeutic Global Market to 2030 - Multifactorial Disease Targeting Presents Opportunities

Global RNA Based Therapeutics Market
Dublin, April 28, 2022 (GLOBE NEWSWIRE) -- The "RNA Based Therapeutic Market by Disease Type, Application and End User: Global Opportunity Analysis and Industry Forecast, 2021-2030" report has been added to ResearchAndMarkets.com's offering.
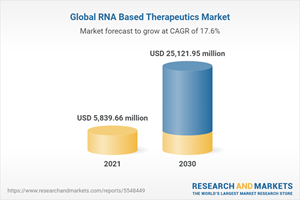
The global RNA based therapeutics market generated $4,938.38 million in 2021, and is projected to reach $25,121.95 million by 2030, growing at a CAGR of 17.6% from 2021 to 2030.
RNA based therapeutics is a class of medications based on ribonucleic acid (RNA). RNA based therapeutics provides or modifies ribonucleic acid (RNA) to patients' cells. Now it has the potential to treat a wide variety of diseases, including cardiovascular disease, hemophilia, and cancer. Based on messenger RNA (mRNA), the main types of RNA therapeutics include antisense RNA (asRNA), RNA interference (RNAi), the agents of RNA interference (RNAi), catalytically active RNA molecules (ribozymes), and RNAs that bind proteins and other molecular ligands (aptamers).
In addition, messenger RNA (mRNA) can be used as protein replacement therapy to treat diseases caused by a lack of protein, or by defective proteins, such as cystic fibrosis. Moreover, Prominent players in the global RNA based therapeutics market have opted various strategies such as type launches, approvals, and investments in R&D for advancement in RNA based therapeutics to strengthen their position in the market and sustain the competitive environment
The global RNA based therapeutics market has garnered significant attention in the recent years due to its potential to treat variety of chronic diseases such as cancer, diabetes, Human immunodeficiency virus infection and acquired immune deficiency syndrome (AIDS), Tuberculosis and certain cardiovascular conditions and rise in government initiatives to promote healthy lifestyle.
Furthermore, rise in health awareness significantly contributes toward the growth of the market. However, factors such as dearth of skilled professionals and end-user budget constraints in developing countries are expected to restrain the market growth. Conversely, surge in number of applications in molecular diagnostics, particularly in cancer and improving synthetic delivery carriers and chemical modifications to RNA is expected to offer profitable opportunities for the market players in the near future. Furthermore, increase in demand for genomic sequencing in laboratories, and research centers, also increasing molecular diagnostics are expected to provide lucrative opportunities for the growth of the market in near future.
RNA based therapeutics is segmented into by type, by application, and by end user. Based on type, the market is segmented into RNA interference (RNAi) technologies and RNA antisense technologies. Based on application, the market is segmented into genetic disorders and auto immune disorders. Based on end user, the market is segmented into research institutes, and hospitals & clinics. Region wise, it is classified into the North America, Europe, Asia-Pacific, and LAMEA.
Key Benefits
The study provides an in-depth analysis of the global RNA based therapeutic market along with the current trends and future estimations to elucidate the imminent investment pockets.
A comprehensive analysis of the factors that drive and restrict the market growth is provided in the report.
Comprehensive quantitative analysis of the industry from 2020 to 2030 is provided to enable the stakeholders to capitalize on the prevailing market opportunities.
Extensive analysis of the key segments of the industry helps to understand the products and end users of RNA based therapeutic used across the globe.
Key market players and their strategies have been analysed to understand the competitive outlook of the market.
Key Topics Covered:
CHAPTER 1- INTRODUTION
CHAPTER 2: EXECUTIVE SUMMARY
CHAPTER 3: MARKET OVERVIEW
3.1. Market definition and scope
3.2. Key findings
3.2.1. Top investment pockets
3.2.2. Top winning strategies
3.3. Top player positioning, 2020
3.4. Porter's five forces analysis
3.4.1. Bargaining power of suppliers
3.4.2. Bargaining power of buyers
3.4.3. Threat of new entrants
3.4.4. Threat of substitutes
3.4.5. Intensity of competitive rivalry
3.5. Market dynamics
3.5.1. Drivers
3.5.1.1. Target Specificity and Selectivity of Treatment
3.5.1.2. Initiatives From Government for Large-Scale Sequencing Projects
3.5.2. Restraints
3.5.2.1. Hurdles In Drug Delivery
3.5.2.2. High Cost of Research and Threat of Failure
3.5.3. Opportunities
3.5.3.1. Early Commercialization of Pipeline Therapeutics Assists to Gain Competitive Advantage
3.5.3.2. Multifactorial Disease Targeting
3.5.4. Impact Analysis
3.1. COVID-19 impact analysis on the RNA based therapeutics market
CHAPTER 4: RNA BASED THERAPEUTICS MARKET, BY TYPE
4.1. Overview
4.1.1. Market size and forecast, by Type
4.2. RNA Interference (RNAi)
4.2.1. Key market trends, growth factors, and opportunities
4.2.2. Market size and forecast
4.2.3. Market analysis, by country
4.3. RNA Antisense
4.3.1. Key market trends, growth factors, and opportunities
4.3.2. Market size and forecast
4.3.3. Market analysis, by country
CHAPTER 5: RNA BASED THERAPEUTICS MARKET, BY APPLICATION
5.1. Overview
5.1.1. Market size and forecast, by end user
5.2. Genetic Disorders
5.2.1. Market size and forecast
5.2.2. Market analysis, by country
5.3. Auto immune disorders
5.3.1. Market size and forecast
5.3.2. Market analysis, by country
CHAPTER 6: RNA BASED THERAPEUTICS MARKET, BY END USER
6.1. Overview
6.1.1. Market size and forecast, by end user
6.2. Research Institutes
6.2.1. Market size and forecast
6.2.2. Market analysis, by country
6.3. Hospitals & Clinics
6.3.1. Market size and forecast
6.3.2. Market analysis, by country
CHAPTER 7: RNA BASED THERAPEUTICS MARKET, BY REGION
CHAPTER 8: COMPANY PROFILES
8.1. Alnylam Pharmaceuticals Inc
8.1.1. Company overview
8.1.2. Company snapshot
8.1.3. Operating business segments
8.1.4. Product portfolio
8.1.5. Business performance
8.1.6. Key strategic moves and developments
8.2. Arbutus Biopharma Corporation
8.2.1. Company overview
8.2.2. Company snapshot
8.2.3. Operating business segments
8.2.4. Product portfolio
8.2.5. Business performance
8.3. Arrowhead Pharmaceuticals
8.3.1. Company overview
8.3.2. Company snapshot
8.3.3. Operating business segments
8.3.4. Product portfolio
8.3.5. Business performance
8.4. Benitec Biopharma Inc.
8.4.1. Company overview
8.4.2. Company snapshot
8.4.3. Operating business segments
8.4.4. Product portfolio
8.4.5. Business performance
8.5. Biogen
8.5.1. Company overview
8.5.2. Company snapshot
8.5.3. Operating business segments
8.5.4. Product portfolio
8.5.5. Business performance
8.6. Gradalis, Inc.
8.6.1. Company overview
8.6.2. Company snapshot
8.6.3. Operating business segment
8.6.4. Product portfolio
8.7. Genzyme (Sanofi), Inc.
8.7.1. Company overview
8.7.2. Company snapshot
8.7.3. Operating business segments
8.7.4. Product portfolio
8.7.5. Business performance
8.7.6. Key strategic moves and developments
8.8. Ionis Pharmaceuticals, Inc.
8.8.1. Company overview
8.8.2. Company snapshot
8.8.3. Operating business segments
8.8.4. Product portfolio
8.8.5. Business performance
8.9. Sarepta Therapeutics
8.9.1. Company overview
8.9.2. Company snapshot
8.9.3. Operating business segments
8.9.4. Product portfolio
8.9.5. Business performance
8.9.6. Key strategic moves and developments
8.10. Silence Therapeutics plc
8.10.1. Company overview
8.10.2. Company snapshot
8.10.3. Operating business segments
8.10.4. Product portfolio
For more information about this report visit https://www.researchandmarkets.com/r/5j6045
Attachment
CONTACT: CONTACT: ResearchAndMarkets.com Laura Wood, Senior Press Manager press@researchandmarkets.com For E.S.T Office Hours Call 1-917-300-0470 For U.S./CAN Toll Free Call 1-800-526-8630 For GMT Office Hours Call +353-1-416-8900


 Yahoo Finance
Yahoo Finance 
