QIAGEN (QGEN) to Join DAX Riding on Molecular Diagnostics Feat
Netherlands-based QIAGEN N.V. QGEN is set to join the coveted broader market index, DAX (Deutscher Aktienindex) on Sep 20. Being selected to be part of the leading stock market index in Germany can be considered a major milestone for QIAGEN.
QIAGEN got the opportunity to enter this prestigious index following DAX’s recent expansion. The index will now include 40 major Germany-based blue-chip companies compared with 30 stocks earlier. This change will be effective from Sep 20 itself. This is also the date when QIAGEN will exclude itself from Germany’s MDAX, which will now be the index for the next 50 companies based on market capitalization after the DAX.
This sample and assay technologies player with its operational headquarters in both the United States and Germany came into the limelight on its growing potential in the molecular diagnostics testing space worldwide. Further, through the months of the pandemic, the company’s business has expanded significantly in the field of COVID-19 testing.
According to QIAGEN, joining the ranks of Germany’s top publicly-listed companies is a great achievement itself.
QIAGEN continues to be a member of Germany’s TecDAX that comprises the 30 largest Germany-based companies from the technology sector. Being listed under the New York Stock Exchange (NYSE) as well, it is one of the few companies trading on exchanges in both countries.
Inflating Market Cap in the Recent Years
This global provider of sample and assay technologies for molecular diagnostics, applied testing, academic and pharmaceutical research, ended 2019 with a market capitalization of $7.69 billion. This increased by 57.1% in 2020 to $12.08 billion.
QGEN's Strides in COVID and Non-COVID Lines of Research
Through 2020, QIAGEN launched several COVID-19 solutions. The company, in August 2020, initiated the U.S. launch of the new QIAreach Anti-SARS-CoV-2 Total Test, an easy-to-use digital test done on a portable device to detect antibodies in people exposed to the SARS-CoV-2 virus. Also, it launched the QIAseq SARS-CoV-2 Primer Panel for NGS of the coronavirus genome along with integrated analysis and interpretation workflows for insights into the evolution and spread of the virus.
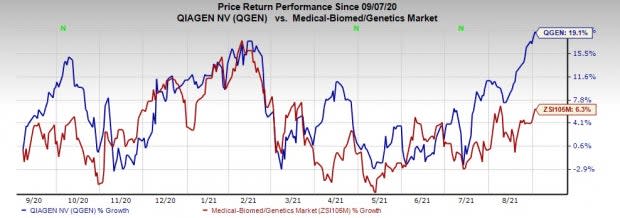
Image Source: Zacks Investment Research
In November 2020, the company announced the European launch of the NeuMoDx Flu A-B/RSV/SARS-CoV-2 Vantage Test to identify influenzas A and B, respiratory syncytial virus (RSV), and SARS-CoV-2 infections. Further, QIAGEN also commercialized a portable digital test — QIAreach SARS-CoV-2 Antigen Test — in the United States that can be used by laboratories to detect SARS-CoV-2 antigens in people with active infections within a lesser time period.
In May 2021, QIAGEN received the FDA’s Emergency Use Authorization (EUA) for QIAreach Anti-SARS-CoV-2 Total Test (that processes up to 32 swab samples per hour), developed in partnership with Australia’sdigital diagnostics company, Ellume.
QIAGEN has also experienced further improvements in non-COVID areas of the portfolio.
The company recorded robust demand for sample preparation instruments through 2020, thus enabling the company to place over 400 new placements of the QIAsymphony automation system in 2020, reflecting a 50% increase over 2019. This marked total cumulative placements in more than 2,900 systems. In the QIAcube family of instruments, more than 9,800 cumulative placements were made at the end of 2020, building on more than 8,000 placements at the end of 2019. Further, over 400 EZ1 instruments were placed in 2020, indicating a surge of about 60% in annual placements and building on over 3,750 cumulative placements at the end of 2019.
In 2021, the company launched QIAcube Connect for molecular diagnostic and the new QIAprep& kit. Further, QIAGEN’s U.S. test menu expanded with the receipt of the EUA for a four-plex test, with simultaneous detection of flu A and B, RSV, and SARS-CoV-2. The company is also progressing with its QuantiFERON franchise with the recent release of the LymeDetect assay on DiaSorin LIAISON platform. It also launched the FDA-approved tissue companion diagnostic -- therascreen KRAS RGQ PCR Kit to help in the detection of non-small cell lung cancer (NSCLC) patients that may be eligible for treatment with LUMAKRAS (sotorasib).
Price Performance
Shares of the company have gained 19.1% in a year’s time compared with the industry’s rise of 6.3%.
Zacks Rank and Key Picks
Currently, QIAGEN carries a Zacks Rank #3 (Hold).
A few better-ranked stocks from the broader medical space are Envista Holdings Corporation NVST, BellRing Brands, Inc. BRBR,and Henry Schein, Inc. HSIC, each carrying a Zacks Rank #2 (Buy). You can see the complete list of Zacks #1 Rank (Strong Buy) stocks here.
Envista Holdings has an estimated long-term earnings growth rate of 27%.
BellRing Brands has an estimated long-term earnings growth rate of 29%.
Henry Schein has a projected long-term earnings growth rate of 14%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Henry Schein, Inc. (HSIC) : Free Stock Analysis Report
QIAGEN N.V. (QGEN) : Free Stock Analysis Report
Envista Holdings Corporation (NVST) : Free Stock Analysis Report
BellRing Brands, Inc. (BRBR) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 