Atea Pharma (AVIR) Gains on Upbeat Initial Data From HCV Study
Atea Pharmaceuticals, Inc. AVIR, a clinical-stage company, reported positive initial data from the first 52 patients in the lead-in cohort of the mid-stage study evaluating its bemnifosbuvir and ruzasvir (RZR) combination therapy for the treatment of Hepatitis C Virus (HCV). Bemnifosbuvir is AVIR’s nucleotide polymerase inhibitor, while RZR is an oral NS5A inhibitor.
The lead-in cohort of the phase II study enrolled 60 non-cirrhotic patients previously untreated with direct-acting antivirals, across all genotypes. These patients were administered 550 mg of bemnifosbuvir in combination with 180 mg of RZR once daily for eight weeks. The preliminary data readout is from 52 of the 60 lead-in patients after four weeks of treatment.
Sustained Virologic Response at Week 4 (SVR4) is the decision endpoint of the mid-stage study, the purpose of which is to substantially shorten the anticipated timeline for the completion of the study. The continuation of the study is contingent upon the fulfillment of this decision endpoint.
Initial results for the first 52 patients observed an SVR4 rate of 98%, which exceeded Atea Pharma’s efficacy criterion of >90% for continuing the study. The data included one patient with poor adherence who did not achieve SVR4.
The combination demonstrated a favorable safety profile and was overall well tolerated. Adverse events in the study were mostly mild in severity with no treatment discontinuations.
Based on these findings, Atea Pharma is gearing up to complete enrolling patients in the phase II study. The company anticipates enrolling 220 additional patients across all genotypes in the phase II study, which is expected to be completed by mid-2024.
The phase II study is currently evaluating the safety and efficacy of eight weeks of treatment with the bemnifosbuvir/RZR combo therapy in treatment-naïve HCV-infected patients, either without cirrhosis and/or with compensated cirrhosis. The primary endpoint of the study is the Sustained Virologic Response at Week 12 (SVR12).
Management states that it has been observed in clinical studies of other direct-acting antiviral therapy combinations that the SVR4 result is highly correlated with SVR12.
Top-line results from the mid-stage study of the bemnifosbuvir/RZR combo for HCV are expected in the third quarter of 2024.
Atea Pharma’s stock gained 11.3% in the last trading session as the investors cheered the encouraging update from the phase II HCV study. Over the past year, shares of AVIR have plunged 28.1% compared with the industry’s 11.4% decline.
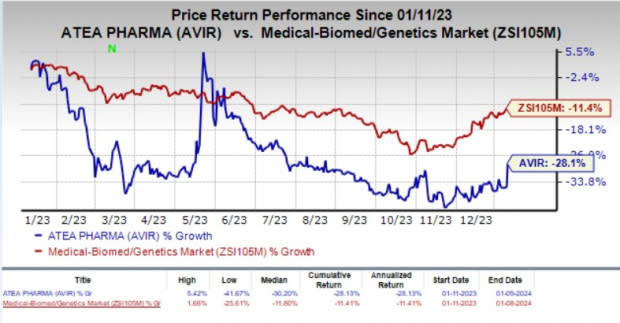
Image Source: Zacks Investment Research
Per the company, there are approximately 2.4 million people in the United States infected with HCV. Despite the transformative effect of currently available direct-acting antivirals in HCV treatment, the rate of new and reinfection exceeds cure rates in the United States. This represents a significant unmet medical need.
In the same press release, Atea Pharma also reported the achievement of a significant enrollment milestone in its late-stage COVID-19 study, which could also have contributed to the stock price gain.
Per the company, more than 650 patients were enrolled in the monotherapy arm of its phase III SUNRISE-3 study for the treatment of COVID-19. Further enrollment also continues with the current winter wave.
The achievement of this enrollment milestone allows for the first interim analysis of the late-stage study for the safety and futility of the investigational candidate by an independent data monitoring committee. The results of the interim analysis are expected in March 2024.
The phase III SUNRISE-3 registrational study is evaluating bemnifosbuvir or placebo administered concurrently with locally available standard-of-care treatments for high-risk COVID-19 patients. Patients enrolled in the study are equally divided to receive either a twice-daily 550 mg dose of bemnifosbuvir or placebo twice daily for five days. The primary endpoint of the study is all-cause hospitalization or death through Day 29 post-treatment in the monotherapy arm in 2,200 patients.
Atea Pharmaceuticals, Inc. Price and Consensus
Atea Pharmaceuticals, Inc. price-consensus-chart | Atea Pharmaceuticals, Inc. Quote
Zacks Rank & Stocks to Consider
Atea Pharma currently has a Zacks Rank #3 (Hold).
Some better-ranked drug/biotech stocks are Puma Biotechnology, Inc. PBYI, ADMA Biologics ADMA and Acadia Pharmaceuticals ACAD. While PBYI sports a Zacks Rank #1 (Strong Buy), ADMA and ACAD carry a Zacks Rank #2 (Buy) each at present. You can see the complete list of today’s Zacks #1 Rank stocks here.
In the past 30 days, the Zacks Consensus Estimate for Puma Biotech’s 2023 earnings per share (EPS) has increased from 72 cents to 73 cents. During the same time frame, the consensus estimate for Puma Biotech’s 2024 EPS has increased from 64 cents to 69 cents. Over the past year, shares of PBYI have gained 0.4%.
PBYI beat estimates in three of the last four quarters while missing on one occasion, delivering a four-quarter average earnings surprise of 76.55%.
In the past 30 days, the Zacks Consensus Estimate for ADMA Biologics’ 2023 loss per share has narrowed from 3 cents to 2 cents. The consensus estimate for ADMA Biologics’ 2024 EPS is pegged at 20 cents. Over the past year, shares of ADMA have gained 38.9%.
ADMA beat estimates in three of the trailing four quarters and matched in one, delivering an average earnings surprise of 63.57%.
In the past 30 days, the Zacks Consensus Estimate for Acadia’s 2023 loss per share has remained constant at 33 cents. During the same time frame, the consensus estimate for Acadia’s 2024 EPS is pegged at $1.04. Over the past year, shares of ACAD have rallied 77.2%.
ACAD beat estimates in two of the trailing four quarters and missed the mark on the other two occasions, delivering an average earnings surprise of 20.69%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Puma Biotechnology, Inc. (PBYI) : Free Stock Analysis Report
ADMA Biologics Inc (ADMA) : Free Stock Analysis Report
ACADIA Pharmaceuticals Inc. (ACAD) : Free Stock Analysis Report
Atea Pharmaceuticals, Inc. (AVIR) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 