Corcept (CORT) Up YTD on Robust Korlym Sales, Pipeline Advances
Shares of Corcept Therapeutics Incorporated CORT have rallied 34.8% so far this year against the industry’s decline of 30.1%.
The company’s sole marketed drug, Korlym, is approved for the once-daily oral treatment of hyperglycemia — secondary to hypercortisolism — in adult patients with endogenous Cushing’s syndrome who are suffering from type II diabetes or glucose intolerance and have already failed surgery or are unsuitable for the same.
CORT’s top line solely comprises Korlym sales, which witnessed a year-over-year increase during the first nine months of 2022. The drug generated sales worth $298.8 million in the first nine months of 2022, reflecting a year-over-year increase of 11.8%. Korlym has been a consistent revenue driver since its approval.
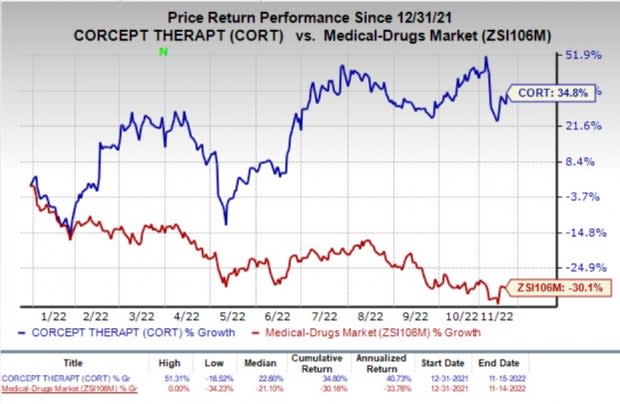
Image Source: Zacks Investment Research
Corcept's other pipeline candidates are also progressing with several data readouts, expected later this year and next year.
CORT’s lead pipeline candidate, relacorilant, is being evaluated in phase III of the GRACE study to treat Cushing’s syndrome. Enrollment is currently ongoing in said study. A new drug application for relacorilant is expected to be submitted in the second half of 2023.
Relacorilant is also being investigated in the phase III of the GRADIENT study in patients, whose Cushing’s syndrome is caused by adrenal adenoma. Enrollment in this study is ongoing.
Corcept is also evaluating relacorilant in combination studies for treating solid tumors. The pivotal phase III ROSELLA study is evaluating relacorilant in combination with nab-paclitaxel for the treatment of patients with platinum-resistant ovarian cancer.
A phase Ib study is evaluating relacorilant in combination with Merck’s MRK blockbuster PD-1 checkpoint inhibitor, Keytruda (pembrolizumab), for treating patients suffering from adrenal cancer along with cortisol excess.
Merck’s biggest revenue generator, Keytruda, is approved for treating several types of cancer indications. MRK continues to study Keytruda for addressing more cancer indications.
Successful development of relacorilant for additional indications will be an added boost to the company.
Last month, CORT initiated the phase II DAZALS study evaluating its pipeline candidate, dazucorilant, for treating patients with amyotrophic lateral sclerosis, a destructive neuromuscular illness.
The company is evaluating miricorilant in the phase II GRATITUDE study for treating the reversal of antipsychotic-induced weight gain (“AIWG”). The phase II GRATITUDE 2 study is investigating miricorilant to reverse long-standing AIWG. Data from both studies are expected later in 2022.
CORT is conducting a phase Ib dose-finding study evaluating miricorilant in patients with presumed non-alcoholic steatohepatitis. Data from the same is also expected in the first half of 2023.
The successful development and potential approval of other candidates are likely to lend a boost to Corcept’s stock and should lower its dependence on Korlym.
However, the company remains heavily dependent on Korlym for growth. Moreover, its pipeline is still some time away from commercialization and generating incremental sales. Hence, a decline in Korlym sales is likely to significantly hurt the company’s prospects.
Corcept Therapeutics Incorporated Price
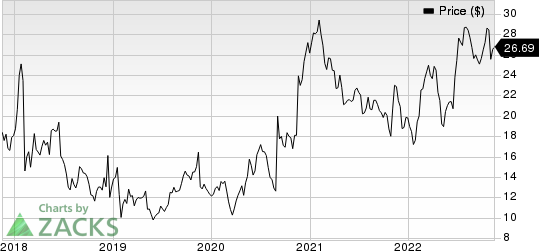
Corcept Therapeutics Incorporated price | Corcept Therapeutics Incorporated Quote
Zacks Rank & Stocks to Consider
Corcept currently carries a Zacks Rank #3 (Hold). Some better-ranked stocks in the same sector are Catalyst Pharmaceuticals, Inc. CPRX and United Therapeutics Corporation UTHR, both carrying a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Earnings estimates for Catalyst Pharmaceuticals have been revised upward by 1.4% for 2022 and 1.2% for 2023 in the past 60 days.
Earnings of Catalyst Pharmaceuticals surpassed estimates in two of the trailing four quarters and missed on two occasions. CPRX delivered a negative earnings surprise of 4.10% on average.
Earnings estimates for United Therapeutics have been revised upward by 9.8% for 2022 and 8.3% for 2023 in the past 60 days.
Earnings of United Therapeutics surpassed estimates in two of the trailing four quarters and missed on the remaining two occasions. UTHR delivered an earnings surprise of 7.24% on average.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Merck & Co., Inc. (MRK) : Free Stock Analysis Report
United Therapeutics Corporation (UTHR) : Free Stock Analysis Report
Corcept Therapeutics Incorporated (CORT) : Free Stock Analysis Report
Catalyst Pharmaceuticals, Inc. (CPRX) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 