Esperion (ESPR) Reports Positive Data From CLEAR Study
Esperion Therapeutics, Inc. ESPR announced that the landmark Cholesterol Lowering via Bempedoic acid, an ACL-Inhibiting Regimen (CLEAR) Outcomes trial of Nexletol (bempedoic acid) met its primary endpoint.
Shares of the company were up following the announcement, but the gains were pared later on Dec 7.
The CLEAR Outcomes is a phase III, event-driven, randomized, multicenter, double-blind, placebo-controlled trial designed to evaluate whether treatment with bempedoic acid reduces the risk of cardiovascular events in patients with or at high risk for cardiovascular disease with documented statin intolerance (inability to tolerate two or more statins, one at a low dose) and elevated LDL-cholesterol levels (fasting blood LDL-C ≥ 100 (2.6 mmol/L)).
Please note that Nexletol is indicated as an adjunct to diet and maximally tolerated statin therapy for the treatment of adults with heterozygous familial hypercholesterolemia or established atherosclerotic cardiovascular disease who require additional lowering of LDL-C.
Data showed a statistically significant reduction in the risk of major adverse cardiovascular events (MACE-4) in patients treated with 180 mg/day of Nexletol compared to placebo.
Shares of Esperion have surged 28.6% so far this year against the industry’s 30.1% decline.
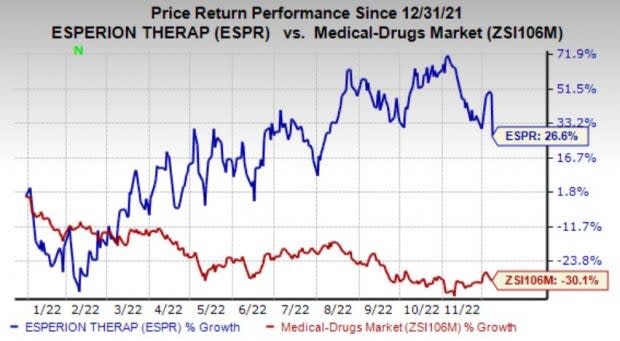
Image Source: Zacks Investment Research
Per the company, bempedoic acid becomes the first ATP-citrate lyase inhibitor to demonstrate significant and clinically meaningful outcomes for patients in whom existing lipid-lowering therapies fall short following these positive top-line results.
The data from this trial further strengthens the clinical evidence supporting the role of bempedoic acid for patients and will boost the growth potential of the drug.
Esperion has two FDA-approved drugs — Nexletol and Nexlizet — in its commercial portfolio that are approved for treating elevated LDL-C (bad cholesterol). These two drugs are marketed as Nilemdo and Nustendi in Europe and several other ex-U.S. markets in partnership with Daiichi Sankyo. Esperion records royalties on sales of its drugs in Europe.
The company plans to submit data to the appropriate regulatory authorities in 2023.
The encouraging uptake of both drugs since their approval in the United States and Europe in 2020 bodes well for the company.
Zacks Rank & Other Stock to Consider
Esperion currently has a Zacks Rank #2 (Buy).
Some other top-ranked stocks in the overall healthcare sector are Gilead Sciences, Inc. GILD, Biogen BIIB and Vertex Pharmaceuticals VRTX. All three carry the same rank as Esperion. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Earnings estimates for Gilead Sciences have increased 48 cents in the last 60 days to $7.09. Gilead has surpassed earnings estimates in three of the past four quarters with an average positive beat of 0.36%.
Earnings estimates for Vertex Pharmaceuticals have increased 44 cents in the last 60 days to $14.65. Vertex has surpassed earnings estimates in three of the past four quarters with an average positive beat of 3.16%.
Earnings estimates for Biogen have increased 55 cents in the last 60 days to $16.98. Biogen has surpassed earnings estimates in three of the past four quarters with an average positive beat of 7.02%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Biogen Inc. (BIIB) : Free Stock Analysis Report
Gilead Sciences, Inc. (GILD) : Free Stock Analysis Report
Vertex Pharmaceuticals Incorporated (VRTX) : Free Stock Analysis Report
Esperion Therapeutics, Inc. (ESPR) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 