Global Bioprocess Validation Market Report 2023: Rising Usage of Single Use Bioprocessing Drives Growth

Global Bioprocess Validation Market
Dublin, March 15, 2023 (GLOBE NEWSWIRE) -- The "Global Bioprocess Validation Market Size, Share & Industry Trends Analysis Report By Stage, By Mode, By Testing Type, By Regional Outlook and Forecast, 2022-2028" report has been added to ResearchAndMarkets.com's offering.
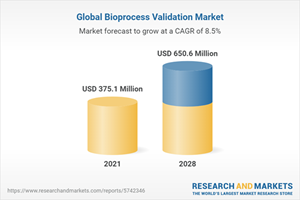
The Global Bioprocess Validation Market size is expected to reach $650.6 Million by 2028, rising at a market growth of 8.5% CAGR during the forecast period.
The documentation of all procedures, evidence, and activities of the process of biological and biopharmaceutical product formation is known as bioprocess validation. The documentation is performed as per the guidelines of various regulatory bodies, for instance, US FDA (Food and Drug Administration).
It ensures the maintenance of compliance in all the stages of the product testing procedures. Assessing Active Pharmaceutical Ingredients (API) and impurities is essential to bioprocess validation. The primary principle of validation is to ensure that every process of the bioproduct processing is evaluated, which provides quality products, maintaining scientifically documented evidence.
Quality, efficacy, and safety are maintained during the bioprocess validation. Validation has become an essential issue in manufacturing biopharmaceuticals or biologies intended for therapeutic usage. In addition to the validation of final product quality, manufacturing process validation is gaining wide attention.
Bioprocesses are sensitive and delicate and hence require careful planning and organization for successful process validation. As a result, bioprocess chromatography is the most generally utilized unit operation in biologics manufacturing.
Market Growth Factors
The rise in the need to outsource bioprocess validation
In the pharmaceutical sector, compliance with the standards of different regulatory bodies is essential, which is why bioprocess validation is an integrated process in pharmaceuticals. Validation verifies that all procedures stick to the stated requirements by the government's regulations. Any validation method requires exhaustive documentation that conforms to standard operating procedures and ongoing activities.
Pharmaceutical manufacturing is being outsourced to third-party service providers to raise production yields. The wide usage of disposable technologies in medication development aids in reducing production costs. The increasing outsourcing of bioprocesses validation to third-party service providers will propel the growth of the bioprocess validation market.
Rising usage of single use bioprocessing
The implementation of single-use bioprocessing systems enhances manufacturing processes' productivity by reducing automation's cost and complexity. It also eliminates the requirement for changeover cleaning/validation between consecutive operations. In addition, single-use bioprocessing systems eliminate the need for additional investments by removing the requirement for sterilization.
The initial investment costs with single-use bioprocessing systems are lower compared to that of the stainless-steel facility at the same scale, making it suitable for new players with low investments. Thereby boosting the demand for the bioprocess validation market.
Market Restraining Factor
Possible issues with extractables & leachable
The food and pharmaceutical sectors are dealing with difficulties with the trace quantity of contaminants generated by the extraction or leaching process. Even after the strict regulations of many nations' governments, corporations are still disregarding such issues and the risk to the lives of consumers/patients. Pharmaceutical manufacturers and regulatory agencies are concerned with extractable and leachable. Any containers and pharmaceutical packaging system, including plastic bottles, labeling ink, packing materials, glass, and foil pouches, can leach undesired pollutants into the food ingredient or drug products. Oral drugs, parental and ophthalmic products often pose a more eminent danger. Hence, the issues related to extractables and leachable are expected to hamper the growth of the bioprocess validation market.
Testing Type Outlook
Based on testing type, the bioprocess validation market is segmented into extractables & leachables testing, bioprocess residuals testing, viral clearance testing, filtration & fermentation systems testing and others. The extractables & leachables testing segment garnered a significant revenue share in the bioprocess validation market in 2021. The growth is attributed to the presence of good manufacturing practice guidelines and US FDA regulations. Due to this, there is a huge demand for certified quality bioproducts. The biotechnology and biopharmaceutical companies are engaged in manufacturing cGMP-certified bioproducts. The requirement for government-certified products will propel the segment's growth in the forecasted period.
Stage Outlook
On the basis of stage, the bioprocess validation market is divided into process design, process qualification, and continued process verification. The continued process verification segment witnessed the largest revenue share in the bioprocess validation market in 2021. This is because it collects the processing, data collection analysis, and storage of every batch.
The primary reasons for creating a continued process verification plan are to achieve compliance with regulatory bodies, prevent batch discards, and mitigate process vulnerabilities while finding constant enhancement possibilities. These features and the automation in this process will boost the segment's expansion.
Mode Outlook
By mode, the bioprocess validation market is bifurcated into in house and outsourced. The outsourced segment projected a substantial revenue share in the bioprocess validation market in 2021. This is due to biopharmaceutical and biotechnology companies' high demand for testing services. There is an increase in expenditure for healthcare and the supply of raw materials needed by the market players. In addition, there is also a rising number of CDMOs (Contract Development and Manufacturing Organization) providing drug development and manufacturing services to the pharma and biopharma industries.
Regional Outlook
Region-wise, the bioprocess validation market is analyzed across North America, Europe, Asia Pacific, and LAMEA. The North America region dominated the bioprocess validation market with the largest revenue share in 2021. This is because of significant outsourcing services providers in the region, which leads to the rise in life science research and the production of biologics. Also, the region has many FDA-approved biopharmaceuticals and biotechnological industries. The increased government funding for the bioprocess validation process and the conduct of clinical trials, combined with the presence of many key players, will surge the market's growth in the region.
Recent Strategies Deployed in Bioprocess Validation Market
Sep-2022: Lonza collaborated with Touchlight, a biotechnology company. With this collaboration, Lonza would have the capacity to combine a diverse, additional source of DNA in its complete suite for customers developing messenger RNA vaccines and therapeutics and at the same time Touchlight could broaden the channels through which customers can earn access to Lonza's novel doggy bone DNA (dbDNA) technology
Aug-2022: Thermo Fisher Scientific opened a dry powder media manufacturing facility in Grand Island, New York. The new facility would offer the inessential potential to assist the global supply of media and further broadens the site's abilities to deliver the quality technology and materials required for the commercial manufacturing and development of biological and vaccine therapies
Jun-2022: Merck collaborated with Agilent Technologies, a leader in life diagnostics, sciences, and applied chemical markets. Under this collaboration, both companies would advance bioprocess offering with Agilent's leading analytical solutions to provide combined abilities for improved downstream process control and monitoring
Feb-2022-Feb: Sartorius Stedim Biotech, a subgroup of Sartorius, completed the acquisition of the chromatography division of Novasep, a leading supplier of services in the field of purification and molecule production. The acquisition of a chromatography suite consists of chromatography systems suitable for smaller biomolecules and advanced systems for the continuous production of biologics
Scope of the Study
Market Segments Covered in the Report:
By Stage
Continued Process Verification
Process Qualification
Process Design
By Mode
In House
Outsourced
By Testing Type
Bioprocess Residuals Testing
Extractables & Leachables Testing
Viral Clearance Testing
Filtration & Fermentation Systems Testing
Others
By Geography
North America
US
Canada
Mexico
Rest of North America
Europe
Germany
UK
France
Russia
Spain
Italy
Rest of Europe
Asia Pacific
China
Japan
India
South Korea
Singapore
Malaysia
Rest of Asia Pacific
LAMEA
Brazil
Argentina
UAE
Saudi Arabia
South Africa
Nigeria
Rest of LAMEA
Key Market Players
List of Companies Profiled in the Report:
Thermo Fisher Scientific, Inc
Eurofins Scientific SE
Sartorius AG
Merck KGaA
Lonza Group AG
Danaher Corporation
Charles River Laboratories International, Inc
SGS S.A
Labcorp Corporation
Cobetter Filtration equipment Co., Ltd
Key Attributes:
Report Attribute | Details |
No. of Pages | 221 |
Forecast Period | 2021 - 2028 |
Estimated Market Value (USD) in 2021 | $375.1 Million |
Forecasted Market Value (USD) by 2028 | $650.6 Million |
Compound Annual Growth Rate | 8.5% |
Regions Covered | Global |
Key Topics Covered:
Chapter 1. Market Scope & Methodology
Chapter 2. Market Overview
Chapter 3. Strategies Deployed in Bioprocess Validation Market
Chapter 4. Global Bioprocess Validation Market by Stage
Chapter 5. Global Bioprocess Validation Market by Mode
Chapter 6. Global Bioprocess Validation Market by Testing Type
Chapter 7. Global Bioprocess Validation Market by Region
Chapter 8. Company Profiles
For more information about this report visit https://www.researchandmarkets.com/r/ca03ah
About ResearchAndMarkets.com
ResearchAndMarkets.com is the world's leading source for international market research reports and market data. We provide you with the latest data on international and regional markets, key industries, the top companies, new products and the latest trends.
Attachment
CONTACT: CONTACT: ResearchAndMarkets.com Laura Wood,Senior Press Manager press@researchandmarkets.com For E.S.T Office Hours Call 1-917-300-0470 For U.S./ CAN Toll Free Call 1-800-526-8630 For GMT Office Hours Call +353-1-416-8900


 Yahoo Finance
Yahoo Finance 
