Pharmaceutical Industry's Research Outsourcing Drives Demand for Cardiac Safety Services, Fueling Global Market Growth

Global Cardiac Safety Services Market
Dublin, Oct. 23, 2023 (GLOBE NEWSWIRE) -- The "Global Cardiac Safety Services Market by Type (Standalone, Integrated), Services (ECG/Holter Measurement, Blood Pressure Measurement, Cardiac Imaging, Thorough QT Studies), End User (Pharmaceutical & Biopharma, CROs), Region - Forecast to 2028" report has been added to ResearchAndMarkets.com's offering.
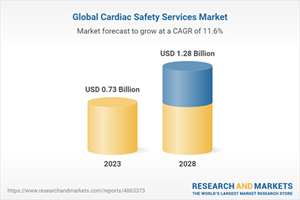
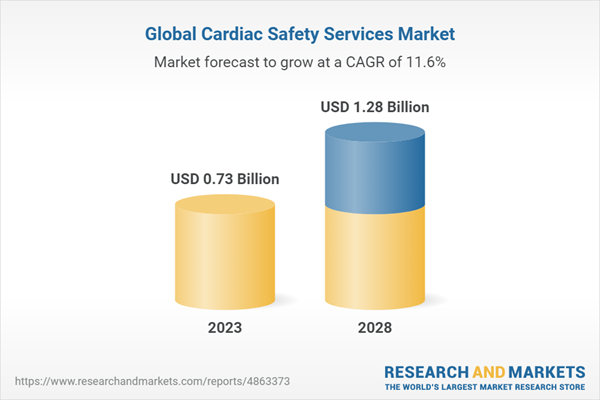
The global cardiac safety services market is on a robust growth trajectory, with projections indicating it will reach USD 1,282 million by 2028, up from USD 739 million in 2023, marking a remarkable CAGR of 11.6% during the forecast period from 2023 to 2028.
The growth of this market can be attributed to several key factors. Firstly, there has been a notable rise in the incidence of cardiovascular diseases, driving the demand for cardiac safety services. Additionally, the pharmaceutical industry is experiencing an upsurge in research and development activities.
Pharmaceutical companies are increasingly outsourcing research tasks to academic organizations and Contract Research Organizations (CROs) to maintain their competitive edge and focus on core functions. This includes outsourcing basic research, genetic engineering, safety studies in animal models, and clinical trials, which in turn has contributed to the high demand for cardiac safety services.
Notably, the pharmaceutical and biopharmaceutical companies segment has been the largest end user of cardiac safety services during the forecast period. These companies utilize these services to oversee clinical trials for newly developed drugs, as they strive to innovate and develop treatments for various medical conditions and diseases. Precise data related to drug development is crucial for their regulatory submissions. Cardiac safety services play a pivotal role in ensuring the safety and efficacy of these drugs, thereby fulfilling regulatory requirements.
In 2022, the pharmaceutical & biopharmaceutical companies segment accounted for the largest share by end user in the global cardiac safety services market, and this trend is expected to continue.
Europe emerges as the second-largest market for cardiac safety services, following North America. The European market has seen substantial growth, driven by demographic shifts such as an aging population and a growing incidence of chronic diseases. With more than 20% of Europeans projected to be aged 65 and above by 2025, there is a strong demand for healthcare services, leading to increased clinical trial activity in pharmaceutical and biopharmaceutical companies. This, in turn, bolsters the cardiac safety services market in the region.
Key Highlights:
Rising Incidence of Cardiovascular Diseases to Drive Market Growth During Forecast Period.
Integrated Services Segment Accounted for Largest Share of North American Market in 2022.
Ecg/Holter Measurement Services Segment to Dominate Market During Forecast Period.
European Countries to Witness Highest Growth Rate During Forecast Period.
Market Dynamics:
Drivers:
Rising Incidence of Cardiovascular Diseases.
Increasing Outsourcing of Services to CROs.
High Number of Clinical Trials for Cardiac Treatments.
Restraints:
Stringent Regulations and Compliance Guidelines.
Inadequacy of Cardiotoxicity Testing.
Opportunities:
Emergence of Innovative Testing Methods.
Growth in Biosimilars and Biologics Development.
Challenges:
High Cost of Cardiac Safety Evaluation Services.
Trends:
Rising Technological Advancements.
Transition to Patient-Centric Approach.
Companies Mentioned:
Acm Global Laboratories
Banook Group
Biobeat
Biotrial
Celerion
Certara
Charles River Laboratories
Clario
Crs. Experts. Early Phase.
Eurofins Scientific
Frontage Labs
Icon plc
Iqvia
Koninklijke Philips N.V.
Laboratory Corporation of America Holdings
Medpace
Ncardia
Nova Research Laboratories LLC
Physiostim
Ppd, Inc. (Part of Thermo Fisher Scientific, Inc.)
Richmond Pharmacology
Sgs S.A.
Shanghai Medicilon Inc.
Worldwide Clinical Trials
Wuxi Apptec
Key Attributes:
Report Attribute | Details |
No. of Pages | 199 |
Forecast Period | 2023 - 2028 |
Estimated Market Value (USD) in 2023 | $0.73 Billion |
Forecasted Market Value (USD) by 2028 | $1.28 Billion |
Compound Annual Growth Rate | 11.6% |
Regions Covered | Global |
For more information about this report visit https://www.researchandmarkets.com/r/fkle1a
About ResearchAndMarkets.com
ResearchAndMarkets.com is the world's leading source for international market research reports and market data. We provide you with the latest data on international and regional markets, key industries, the top companies, new products and the latest trends.
Attachment
CONTACT: CONTACT: ResearchAndMarkets.com Laura Wood,Senior Press Manager press@researchandmarkets.com For E.S.T Office Hours Call 1-917-300-0470 For U.S./ CAN Toll Free Call 1-800-526-8630 For GMT Office Hours Call +353-1-416-8900


 Yahoo Finance
Yahoo Finance 
