Prothena (PRTA) Earns Milestone Payment From Novo Nordisk
Prothena Corporation plc PRTA announced that it has earned a $40 million milestone payment from partner Novo Nordisk NVO.
The payment is related to the advancement of NNC6019 (formerly PRX004) in a phase II study for the treatment of ATTR cardiomyopathy.
In July 2021, Novo Nordisk gained full worldwide rights to Prothena’s intellectual property and related rights to its ATTR amyloidosis business and pipeline.
Per the terms, Prothena is eligible to receive up to $1.2 billion upon the achievement of several clinical development and sales milestones, including the $100 million earned to date.
NNC6019 (formerly PRX004) is an investigational antibody designed to deplete amyloid associated with disease pathology in hereditary and wild-type ATTR amyloidosis without affecting the native, normal tetrameric form of the protein.
Prothena had completed a phase I study with PRX004 in patients with hereditary forms of ATTR prior to this acquisition. PRX004 was found to be safe and well tolerated.
In August 2022, Novo Nordisk initiated a phase II study with NNC6019 that is evaluating the safety and efficacy of NNC6019 in 99 people with transthyretin amyloid cardiomyopathy over a period of 52 weeks.
Prothena’s shares have gained 21.6% in the year so far against the industry’s decline of 19.9%.
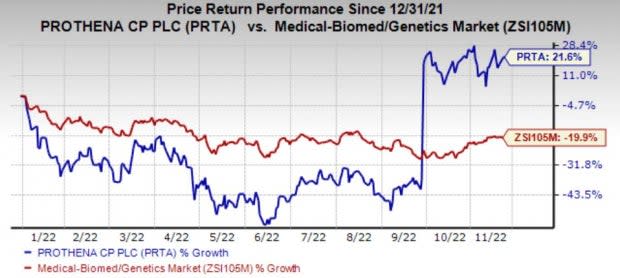
Image Source: Zacks Investment Research
Prothena has been in the spotlight of late due to its promising Alzheimer’s disease (AD) pipeline. In June 2021, the FDA’s approval of Biogen and Eisai’s controversial AD drug, Aduhelm, put the spotlight on companies having promising AD candidates in their pipelines and Prothena was one of them.
Prothena is evaluating PRX012 — an investigational high-potency monoclonal antibody targeting a key epitope at the N-terminus of amyloid beta (Aβ) for treating AD. Prothena is also evaluating PRX005 in collaboration with Bristol Myers BMY.
PRX005 — a potential treatment for AD — is an investigational antibody that targets tau, a protein implicated in diseases, including AD, frontotemporal dementia, progressive supranuclear palsy, chronic traumatic encephalopathy and other tauopathies.
The company is also developing a dual Aβ-Tau vaccine — a potential prevention and treatment for AD — to target key epitopes within Aβ and tau proteins for promoting amyloid clearance and blocking pathogenic tau interaction.
Prothena reported total revenues of $1.5 million and $4.0 million for the third quarter and first nine months of 2022, respectively, primarily from collaboration revenues from BMY.
Prothena currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Bristol Myers Squibb Company (BMY) : Free Stock Analysis Report
Novo Nordisk AS (NVO) : Free Stock Analysis Report
Prothena Corporation plc (PRTA) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 