The Worldwide Anti-Vascular Endothelial Growth Factor Therapeutics Industry is Expected to Reach $13.3 Billion by 2030

Global Anti-Vascular Endothelial Growth Factor Therapeutics Market
Dublin, June 03, 2022 (GLOBE NEWSWIRE) -- The "Anti-Vascular Endothelial Growth Factor Therapeutics Market Share, Size, Trends, Industry Analysis Report, By Product; By Disease; By Region; Segment Forecast, 2022 - 2030" report has been added to ResearchAndMarkets.com's offering.
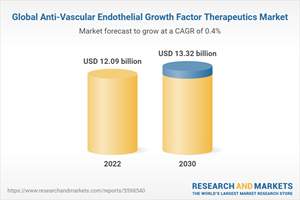
The global anti-vascular endothelial growth factor therapeutics market size is expected to reach USD 13.32 billion by 2030, according to a new study. The report gives a detailed insight into current market dynamics and provides analysis on future market growth.
The anti-vascular endothelial development aspect therapeutics industry is growing due to new product releases, FDA approvals, and research developments. For instance, in September 2021, Byooviz (ranibizumab-nuna) was approved by the US Food and Drug Administration as the first generic drug to Lucentis (ranibizumab injection) for treating a variety of eye diseases and disorders, which include neovascular (wet) age-related macular degeneration (nAMD), a major cause of damage to the optic nerve in Americans aged 65 and older.
Byooviz is also approved for the treatment of macular edoema (fluid build-up) caused by retinal vein obstruction (retinal vein blockage) and myopic choroidal neovascularization, a vision-threatening consequence of myopia (nearsightedness). If approved, faricimab will be the foremost, and only bispecific antibody developed, especially for the eye.
Further, the rising pipeline drugs for the treatment of these diseases, such as AMD, is the aspect that is driving the anti-vascular endothelial development aspect of therapeutics industry development during the forecast period. ADVM-022 and OpRegen are in Phase I and I/II, correspondingly, in the AMD pipeline. Also, GB-102 and KSI-301 are in Phase II, and II/III are in the development phase.
Besides, Amgen, Inc. is exploring its ABP 938, which is in the late stages of development (Phase III). Thus, the pipeline products launched by the major players for the treatment of AMD are the factor boosting the industry development during the forecast period. Based on the product, the eylea segment accounted for the leading share in the industry in 2021. Throughout the forecast period, the category is expected to maintain its dominance due to developments in formulations and sophisticated drug delivery technologies.
Market players such as Bausch Health Companies, Inc., Viatris, Inc., Amgen, Inc., Coherus Biosciences, Eli Lilly, F. Genentech Inc., Hoffmann-La Roche Ltd., Pfizer, Inc., and Xbrane Biopharma AB are some key players operating in the global anti-vascular endothelial growth factor therapeutics industry.
The FDA has also approved the company's diabetic retinopathy application. If agreed, faricimab will be the foremost and singular bispecific antibody explicitly created for the eye. Thus, the product launches and US FDA approvals for the drugs to treat various diseases are the factors boosting the market growth during the forecast period.
Key Topics Covered:
1. Introduction
2. Executive Summary
3. Research Methodology
4. Anti-Vascular Endothelial Growth Factor Therapeutics Market Insights
4.1. Anti-Vascular Endothelial Growth Factor Therapeutics Market - Industry Snapshot
4.2. Anti-Vascular Endothelial Growth Factor Therapeutics Market Dynamics
4.2.1. Drivers and Opportunities
4.2.1.1. Long-term contracts with the partners
4.2.1.2. Rise in the younger population
4.2.2. Restraints and Challenges
4.2.2.1. Data security
4.3. Porter's Five Forces Analysis
4.3.1. Bargaining Power of Suppliers (Moderate)
4.3.2. Threats of New Entrants: (Low)
4.3.3. Bargaining Power of Buyers (Moderate)
4.3.4. Threat of Substitute (Moderate)
4.3.5. Rivalry among existing firms (High)
4.4. PESTLE Analysis
4.5. Anti-Vascular Endothelial Growth Factor Therapeutics Industry trends
4.6. Value Chain Analysis
4.7. COVID-19 Impact Analysis
5. Global Anti-Vascular Endothelial Growth Factor Therapeutics Market, by Product
5.1. Key Findings
5.2. Introduction
5.2.1. Global Anti-Vascular Endothelial Growth Factor Therapeutics, by Product, 2018 - 2030 (USD Billion)
5.3. Eylea
5.3.1. Global Anti-Vascular Endothelial Growth Factor Therapeutics Market, by Eylea, by Region, 2018 - 2030 (USD Billion)
5.4. Lucentis
5.4.1. Global Anti-Vascular Endothelial Growth Factor Therapeutics Market, by Lucentis, by Region, 2018 - 2030 (USD Billion)
5.5. Beovu
5.5.1. Global Anti-Vascular Endothelial Growth Factor Therapeutics Market, by Beovu, by Region, 2018 - 2030 (USD Billion)
6. Global Anti-Vascular Endothelial Growth Factor Therapeutics Market, by Disease
6.1. Key Findings
6.2. Introduction
6.2.1. Global Anti-Vascular Endothelial Growth Factor Therapeutics Market, by Disease, 2018 - 2030 (USD Billion)
6.3. Macular Edema
6.3.1. Global Anti-Vascular Endothelial Growth Factor Therapeutics Market, by Macular Edema, by Region, 2018 - 2030 (USD Billion)
6.4. Diabetic Retinopathy
6.4.1. Global Anti-Vascular Endothelial Growth Factor Therapeutics Market, by Diabetic Retinopathy, by Region, 2018 - 2030 (USD Billion)
6.5. Retinal Vein Occlusion
6.5.1. Global Anti-Vascular Endothelial Growth Factor Therapeutics Market, by Retinal Vein Occlusion, by Region, 2018 - 2030 (USD Billion)
6.6. Age-related Macular Degeneration
6.6.1. Global Anti-Vascular Endothelial Growth Factor Therapeutics Market, by Age-related Macular Degeneration, by Region, 2018 - 2030 (USD Billion)
7. Global Anti-Vascular Endothelial Growth Factor Therapeutics Market, by Geography
8. Competitive Landscape
8.1. Expansion and Acquisition Analysis
8.1.1. Expansion
8.1.2. Acquisitions
8.2. Partnerships/Collaborations/Agreements/Exhibitions
9. Company Profiles
9.1. Amgen, Inc.
9.1.1. Company Overview
9.1.2. Financial Performance
9.1.3. Product Benchmarking
9.1.4. Recent Development
9.2. Bausch Health Companies, Inc.
9.2.1. Company Overview
9.2.2. Financial Performance
9.2.3. Product Benchmarking
9.2.4. Recent Development
9.3. Coherus Biosciences
9.3.1. Company Overview
9.3.2. Financial Performance
9.3.3. Product Benchmarking
9.3.4. Recent Development
9.4. Eli Lilly
9.4.1. Company Overview
9.4.2. Financial Performance
9.4.3. Product Benchmarking
9.4.4. Recent Development
9.5. F. Hoffmann-La Roche Ltd.
9.5.1. Company Overview
9.5.2. Financial Performance
9.5.3. Product Benchmarking
9.5.4. Recent Development
9.6. Genentech Inc.
9.6.1. Company Overview
9.6.2. Financial Performance
9.6.3. Product Benchmarking
9.6.4. Recent Development
9.7. Pfizer, Inc.
9.7.1. Company Overview
9.7.2. Financial Performance
9.7.3. Product Benchmarking
9.7.4. Recent Development
9.8. Viatris, Inc.
9.8.1. Company Overview
9.8.2. Financial Performance
9.8.3. Product Benchmarking
9.8.4. Recent Development
9.9. Xbrane Biopharma AB
9.9.1. Company Overview
9.9.2. Financial Performance
9.9.3. Product Benchmarking
9.9.4. Recent Development
For more information about this report visit https://www.researchandmarkets.com/r/ybcnxb
Attachment
CONTACT: CONTACT: ResearchAndMarkets.com Laura Wood, Senior Press Manager press@researchandmarkets.com For E.S.T Office Hours Call 1-917-300-0470 For U.S./CAN Toll Free Call 1-800-526-8630 For GMT Office Hours Call +353-1-416-8900


 Yahoo Finance
Yahoo Finance 
