Akebia Therapeutics, Inc. (AKBA)
NasdaqCM - NasdaqCM Real-time price. Currency in USD
Add to watchlist
At close: 04:00PM EDT
After hours:
| Previous close | 1.4800 |
| Open | 1.5200 |
| Bid | 1.4200 x 1500 |
| Ask | 1.4400 x 200 |
| Day's range | 1.4100 - 1.5200 |
| 52-week range | 0.7800 - 2.4800 |
| Volume | |
| Avg. volume | 3,243,430 |
| Market cap | 301.818M |
| Beta (5Y monthly) | 0.77 |
| PE ratio (TTM) | N/A |
| EPS (TTM) | N/A |
| Earnings date | N/A |
| Forward dividend & yield | N/A (N/A) |
| Ex-dividend date | N/A |
| 1y target est | N/A |
- Simply Wall St.
Further weakness as Akebia Therapeutics (NASDAQ:AKBA) drops 16% this week, taking five-year losses to 80%
Long term investing is the way to go, but that doesn't mean you should hold every stock forever. We don't wish...
- Simply Wall St.
Revenue Downgrade: Here's What Analysts Forecast For Akebia Therapeutics, Inc. (NASDAQ:AKBA)
The analysts covering Akebia Therapeutics, Inc. ( NASDAQ:AKBA ) delivered a dose of negativity to shareholders today...
- GuruFocus.com
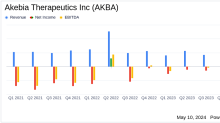
Akebia Therapeutics Reports Q1 2024 Financial Results: A Detailed Overview
Insights into Akebia Therapeutics' Performance and Strategic Developments